Hematological Disorders
Anemia
Anemia is a global health problem. About 25% of the world’s population is anemic. Anemia is highly prevalent in India as well. In 2016, 51% of Indian women in the reproductive age group (15-49 years of age) and 57% of children under the age of 5 years were found to be anemic.
Surprisingly, 25% of Indian men were also found to be anemic. These statistics show that anemia is a national health problem in India.
Anemia Definition: Anemia is defined as a condition in which the hemoglobin concentration is below the normal range, for the age and sex of the individual. In adults, the lower limit of the normal range is taken as 13 g/dL in males and 12 g/dL in females (or hematocrit below 40% in males and 35% in females).
Read and Learn More Pathophysiology
Anemia Symptoms: Subnormal levels of hemoglobin decrease the oxygen-carrying capacity of the blood leading to a deficiency of oxygen in the tissues (hypoxia). The function of tissues with high oxygen demand such as the heart, brain, and exercising muscles is most affected. The symptoms of anemia include
- Pale skin
- Tiredness
- Palpitation
- Easy fatigability
- Generalized muscle weakness
- Lethargy
- Headache
- Light-headedness
- Cold hands and feet
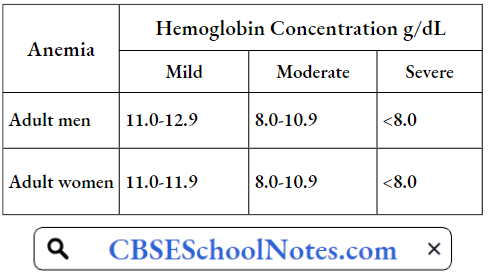
The severity of these symptoms increases with the severity of anemia. WHO has classified anemia as mild, moderate, and severe on the basis of hemoglobin concentration in the blood.
Classification Of Severity Of Anemia
When a patient is diagnosed as suffering from anemia, the treatment depends on its etiology (cause). However, before trying to find the cause, it is helpful to first classify anemia according to the red cell indices (laboratory classification) discussed below.
Red Cell Indices: From the RBC count, hemoglobin concentration, and hematocrit (PCV) value, certain indices (or absolute values) of the red cells of the person can be calculated. These absolute values are used in the laboratory diagnosis of anemia. The method of calculation and normal range of various red cell indices are shown in Table.
On The Basis Of Red Cell Indices, Anemia May Be Classified As
- Microcytic, normocytic or macrocytic, and
- Hypochromic or normochromic.
Once these indices are known, one can proceed to find out the cause.
Calculation And Normal Range Of Red Cell Indices

Aetiological Classification Of Anemias
- Deficiency anemia
- Hemorrhagic anemia
- Haemolytic anemia
- Aplastic anemia
Pathogenesis Of Anemia
1. Deficiency Anemia
Iron Deficiency Anemia: Iron deficiency is the most common cause of anemia in the world. Iron deficiency results in the efficient production of hemoglobin and the number of red blood cells. As explained below, it is more common in females than in males.
- It is important economically because it diminishes the capability of individuals who are affected to perform physical labor, and it diminishes both growth and learning in children. Anemic patients seem to be more prone to infections.
- Iron deficiency anemia is usually the end result of a long period of negative iron balance (iron intake is less than iron excretion). The daily requirement of iron in males is very little. Except for malnourished individuals, males are not prone to developing iron deficiency anemia.
- Therefore, in a male patient with such type of anemia, causes such as gastrointestinal blood loss, malabsorption or hookworm infestation should be looked for.
- Females in the reproductive age group are prone to develop iron deficiency because of loss of iron in menstruation, pregnancy, and lactation. Excessive menstrual losses or repeated pregnancies are the usual causes of iron deficiency anemia in women.
- Gastric surgery and achlorhydria are other causes of iron deficiency anemia which may occur both in males and females.
- Iron deficiency results in the production of a smaller number of red cells, which are not only deficient in hemoglobin (hypochromic) but also smaller in size (microcytic). Thus, in iron deficiency, the MCH is below 26 pg, MCHC below 32%, and MCV below 80 μ3
Severe iron deficiency not only interferes with erythropoiesis but also with cell division in many other tissues. Severe iron deficiency is associated with not only severe anemia but also with disorders of the tongue (atrophic glossitis), esophagus (dysphagia), and nails (koilonychia, spoon-like nails). Iron deficiency can be easily treated by oral administration of Fe2+ salts.

Pernicious Anemia: In India, pernicious anemia is not common. Pernicious anemia is caused by a deficiency of vitamin B12 in the body. Although the vitamin B12 content of the diet of these patients is usually normal, the vitamin is not absorbed in the gut.
- Normally, a glycoprotein (mol. wt. 45,000), known as an intrinsic factor, secreted by the gastric mucosa, helps in the absorption of vitamin B12 in the ileum. Atrophy of gastric mucosa results in the absence of intrinsic factors, leading to malabsorption of vitamin B12.
- Recent evidence suggests that pernicious anemia is an autoimmune disease. The auto-antibodies destroy both the parietal and chief cells of the gastric mucosa (gastric atrophy). Deficiency of vitamin B12 produces a megaloblastic bone marrow reaction and a very severe degree of anemia.
- In the peripheral blood, the red cells are larger in size (macrocytes, MCV greater than 100 μ3) but contain a normal concentration of hemoglobin (normochromic), i.e. vitamin B12 deficiency causes a macrocytic normochromic type of anemia.
The blood smear shows two characteristic features of red cells. Firstly, the cells show a wider variation in shape, i.e. all the red cells are not circular disks and vary in shape (poikilocytosis). Secondly, greater variation of cell size varies (4 pm to 12 pm, average 9.5 um (anisocytosis); normal variation 6.7 to 7.7 pm, average 7.5 pm).
Neural Symptoms In Pernicious Anemia: Vitamin B12 is essential for the synthesis of myelin. Therefore, vitamin B12 deficiency results in the destruction of thick myelinated fibers in the central and peripheral nervous system. Therefore, besides severe anemia, deficiency of vitamin B12 is associated with peripheral neuropathy and degeneration of posterior and lateral white columns of the spinal cord.
Pernicious Anemia Symptoms
- Lack of coordination
- Pain, numbness, and tingling in hands or feet
- Sensory loss
- Weakness of muscles
The disorder, if not treated, is invariably fatal. Pernicious anemia can be treated by regular administration of vitamin B12 by intramuscular route.
Folic Acid Deficiency Anemia: It occurs in individuals who do not take enough folic acid-rich food (green leafy vegetables, fresh fruits, meat). Folic acid deficiency is fairly common during pregnancy. It produces macrocytic normochromic type of anemia but there are no neurological problems. Folic acid deficiency anemia can be easily treated by oral administration of folic acid.
2. Hemorrhagic Anemia: This type of anemia usually results from mild chronic blood loss, for example, due to bleeding piles, excessive menstrual bleeding, or gastric ulcer. Blood loss leads to excessive loss of iron from the body.
Hence such patients usually show hypochromic microcytic (iron deficiency) type of anemia. Treatment of this type of anemia involves oral administration of iron salts as well as treatment of the underlying cause of chronic blood loss.
3. Hemolytic Anemia: The lifespan of normal red blood cells is approximately 120 days. Due to various congenital or acquired defects in the red blood cells, the life span of the red cells may be markedly reduced (as low as 30 days). The bone marrow tries to compensate for the increased rate of red cell destruction by the accelerated rate of erythropoiesis.
- When the rate of red cell regeneration cannot keep pace with the rate of red cell destruction, anemia develops. Moreover, an increased rate of red cell destruction overloads the excretory pathways of hemoglobin degradation products (bilirubin).
- Jaundice (hemolytic type) develops when the rate of bilirubin production exceeds the bilirubin excretory capacity of the liver.
- Hemolytic anemia is usually a normocytic normochromic type since there is no nutritional deficiency. Congenital hemolytic anemia is fairly common in India.
Hemolytic Anemia Causes
- Congenital Hemolytic Anemia
- Congenital (Hereditary) Spherocytosis: This disorder is caused by a congenital defect in the structural proteins in the cell membrane of red cells. Two important proteins, namely, spectrin and ankyrin, maintain the normal shape of red blood cells.
- A genetic defect in the synthesis of either of the two proteins results in a reduced surface area to volume ratio of the red cells. The cells tend to attain a spherical shape. The spherical shape makes the red cells less flexible. Spherocytes cannot bend or twist during passage through narrow capillaries and hence are damaged.
- The spleen seems to possess a special ability to detect and trap even mildly damaged red cells. Greater red cell destruction causes enlargement of the spleen. Thus, besides hemolytic anemia, splenomegaly is an important clinical feature of this disorder. The defect is inherited as an autosomal dominant trait.
- Congenital Disorders Of Hemoglobin: Hemolytic anemia may be due to a congenital defect in the globin chains of hemoglobin. These defects can mainly be divided into
- Hemoglobinopathies when there is an alteration in the amino acid sequences of a polypeptide chain of hemoglobin, for example, Hb S, Fib C, and Fib D.
- The thalassemia in which the amino acid sequence is not disturbed but synthesis of one of the two types of chains (α or β) is impaired.
- In hemoglobinopathies, the altered amino acid sequence results in an abnormality in the solubility of hemoglobin. Consequently, there is a striking abnormality in red cell morphology, which renders the red cell more prone to hemolysis, especially in the spleen.
- In thalassemia, suppression of either α or β chains results in deficient hemoglobin synthesis. Moreover, the absence of one type of polypeptide chain results in excessive production of the other type. This results in a disturbance in hemoglobin solubility and hence excessive hemolysis.
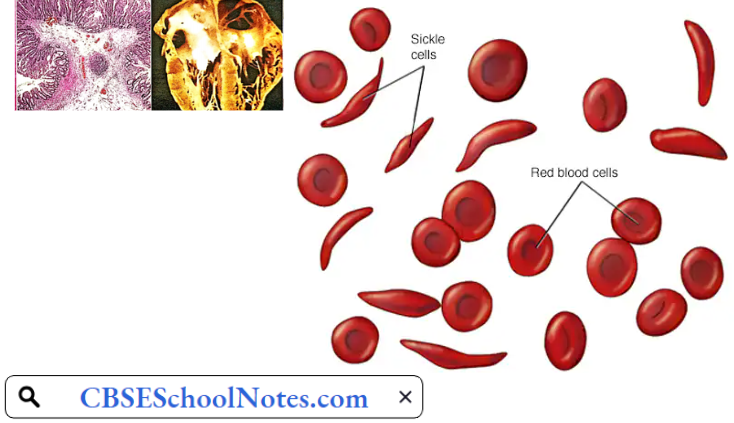
- Sickle Cell Anemia: This hemoglobinopathy is caused by the presence of amino acid valine instead of glutamic acid at position α of the β chains of hemoglobin. This variant is known as haemoglobin S (Hb S) because when deoxygenated, it polymerizes and distorts the red cell membrane into a sickle shape or a crescent shape.
- Hb S is highly prevalent in the black population of Africa but may be found in other countries also. Sickle cell anemia occurs in those individuals who are homozygous for Hb S gene. In such patients, the entire hemoglobin is Hb S type. In individuals who are heterozygous for Hb S gene, the red cells contain Hb S (50%) as well as normal Hb A (50%).
- Such individuals are said to have sickle cell trait. They act as carriers for the abnormal gene but do not suffer from hemolytic anemia.
- The sickle cells are not only abnormal in shape but also less elastic than normal biconcave red cells. The abnormal morphology makes the sickle cells more prone to hemolysis as well as gives a tendency to block the capillaries. The resultant tissue hypoxia causes further sickling of the red cells.
- Thalassemia: In thalassemia, the basic defect is the under-production of one type of the chains of globin component of hemoglobin. Two types of thalassemia are known: Thalassemia α and thalassemia β, depending on the name of the underproduced chains.
- In either case, the condition may be homozygous (thalassemia major) or heterozygous (thalassemia minor). Beta-thalassemia is common in the Mediterranean region, whereas thalassemia is seen in southeast Asia including India. Anemia is mild in patients with thalassemia minor, but very severe in those with thalassemia major.
- In the deficiency of one type of polypeptide chain, the red cells develop the tetramers of the other type, making red cells more prone to hemolysis.
- Congenital (Hereditary) Spherocytosis: This disorder is caused by a congenital defect in the structural proteins in the cell membrane of red cells. Two important proteins, namely, spectrin and ankyrin, maintain the normal shape of red blood cells.
- Acquired Haemolytic Anemia: Acquired hemolytic anemia results from the development of auto-antibodies against the red cells of the patient resulting in excessive destruction of the red cells. The auto-antibodies formed against red cell membrane antigens cause inappropriate destruction of red cells. Such type of anemia is not common in India.
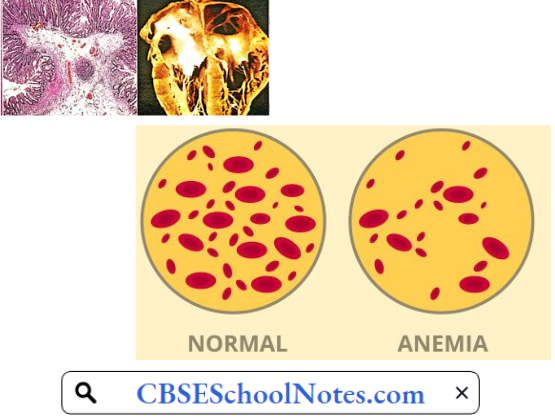
4. Aplastic Anemia: Complete cessation of erythropoiesis is a rare but very serious and often fatal type of anemia. It arises as a complication of hypersensitivity reaction to certain drugs, for example, chloramphenicol, sulfonamides, etc. Excessive irradiation and cytotoxic drugs used in the treatment of malignant disorders also depress the bone marrow.
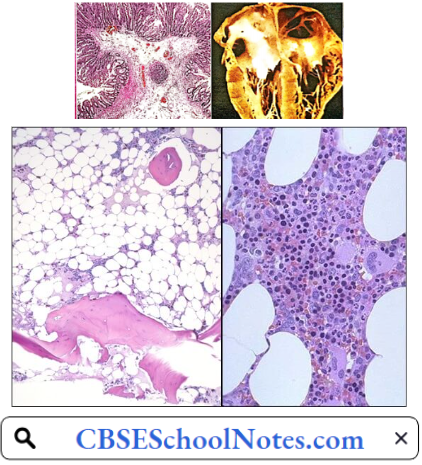
In most of such cases, besides very severe anemia, severe leucopenia, and thrombocytopenia is also present. Death may occur due to infection or severe blood loss. Bone marrow examination reveals the presence of adipose tissue where red bone marrow is normally present indicating cessation of hemopoiesis.
Complications Of Anemia: Left untreated, anemia can cause many health problems, such as
- Severe Fatigue: Severe anemia can make you so tired that you cannot complete everyday tasks.
- Pregnancy Complications: Pregnant women with folate deficiency anemia may be more likely to have complications, such as premature birth.
- Heart problems: Anemia can lead to a rapid or irregular heartbeat (arrhythmia). Severe anemia can lead to an enlarged heart or heart failure.
- Death: Some inherited anemias, such as sickle cell anemia, can lead to life-threatening complications.
Bleeding Disorders
When a blood vessel is injured, hemostasis (stoppage of bleeding) occurs in two stages: Primary hemostasis results in the formation of a platelet plug which stops bleeding temporarily. The next step, secondary hemostasis results in the clotting of blood which stops bleeding permanently.
- Bleeding disorder may be due to a defect in either stage. Accordingly, bleeding disorders can be broadly classified into two categories
- Purpura results from a defect in primary hemostasis.
- Hemophilia results from a congenital defect in secondary hemostasis.
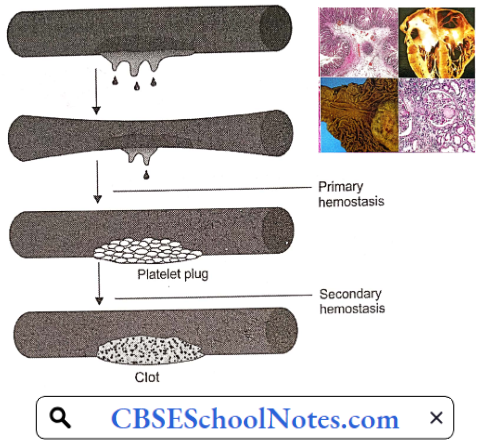
Purpura
Purpura Symptoms: This bleeding disorder is characterized by easy disability and spontaneous multiple hemorrhages under the skin and mucous membranes. At an early stage, the patient may present with numerous red spots of the size of a pinhead on the skin and mucous membranes (petechial hemorrhages). In more severe form, there are larger bleeding spots
- Easy or excessive bruising
- Superficial bleeding into the skin that appears as pinpoint-sized reddish-purple spots (petechiae)
- Bleeding from the gums or nose
- Blood in urine or stools
- Unusually heavy menstrual flow
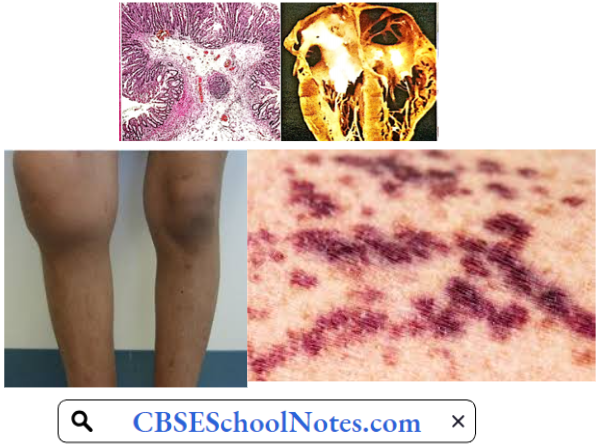
Purpura Aetiology
- Thrombocytopenic purpuras—platelet counts are low.
- Nonthrombocytopenic purpuras—platelet levels are normal, suggesting another cause.
Purpura Pathogenesis: Idiopathic thrombocytopenic purpura (ITP), is an auto-immune disease resulting in greater destruction of platelets in the spleen. The platelet count in the blood is usually less than 50,000/μ3 Decreased platelet count results in a deficiency in the formation of platelet plugs. Hence blood vessels leak blood spontaneously or on mild trauma.
Risk Factors For Purpura Include:
- Infectious diseases, particularly among children and the elderly
- Poor nutrition when it leads to a lack of vitamin C
- Some forms of cancer, such as leukemia and myeloma
- Advanced age
- Poor blood vessel health
Hemophilia
Hemophilia Symptoms: In this disorder, bleeding occurs several hours after an injury. Such bleeding mostly occurs in deep tissues like muscles and joints. Characteristic clinical findings are hemarthrosis (bleeding into a joint) and muscle hematomas.
Hemophilia Aetiology: Hemophilia is a congenital bleeding disorder. Most commonly, there is a congenital deficiency of clotting factor 8. The condition is called hemophilia-A. In a few cases, clotting factor 9 is deficient (hemophilia-B). The disorder is transmitted as an X-chromosome-linked recessive trait. The only effective therapy is intravenous injections of the deficient clotting factor.
Hemophilia Pathogenesis: Factors 8 and 9 are involved in the intrinsic system of coagulation of blood. Deficiency of clotting factor 8 or 9 results in the formation of a weak blood clot in an injured blood vessel. Therefore, the blood vessel starts bleeding again after the effect of primary hemostasis wanes.
Hemophilia Complications
- Deep internal bleeding, for example, deep-muscle bleeding, leads to swelling, numbness or pain of a limb.
- Joint damage from hemarthrosis (hemophilic arthropathy), with severe pain, disfigurement, and even destruction of the joint.
- Intracranial hemorrhage is a serious medical emergency that can cause brain damage and death.
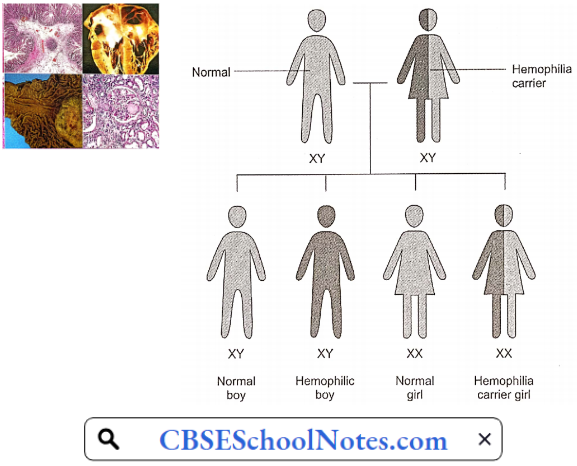
Genetics: The X and Y chromosomes are called sex chromosomes. The gene for hemophilia is carried on the X chromosome. Hemophilia is inherited in an X-chromosome-linked recessive manner. Females inherit two X chromosomes, one from their mother and one from their father (XX).
- Males inherit an X chromosome from their mother and a Y chromosome from their father (XY). That means if a son inherits an X chromosome carrying hemophilia from his mother, he will suffer from hemophilia. It also means that fathers cannot pass hemophilia on to their sons.
- But because daughters have two X chromosomes, even if they inherit the hemophilia gene from their mother, most likely they will inherit a healthy X chromosome from their father and not have hemophilia.
- A daughter who inherits an X chromosome that contains the gene for hemophilia is called a carrier. She can pass the gene on to her children. Hemophilia can occur in daughters but is rare.
For A Female Carrier, There Are Four Possible Outcomes For Each Pregnancy:
- A girl who is not a carrier
- A girl who is a carrier
- A boy without hemophilia
- A boy with hemophilia
Spontaneous mutations of genes account for about 30% of all cases of hemophilia, i.e. family history may be absent.
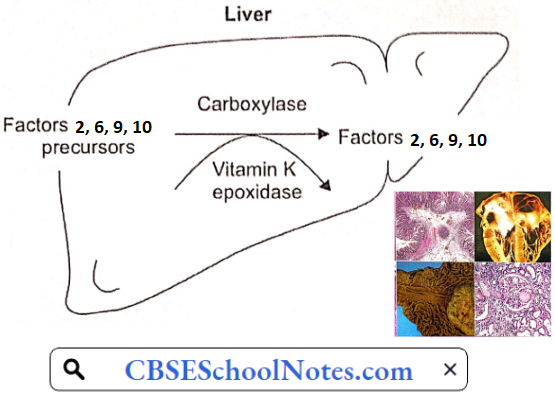
Acquired Defects of Secondary Hemostasis: These defects are far more common than congenital disorders of secondary hemostasis. Clotting factors prothrombin (clotting factor 2) and clotting factors 7, 9, and 10 are synthesized in the liver. Their synthetic reactions require vitamin K.
Severe liver disease or deficiency of vitamin K may cause deficiency of these clotting factors leading to severe and prolonged bleeding after a minor trauma. Intestinal bleeding is one of the serious complications of liver cirrhosis.
Causes Of Vitamin K Deficiency
- Some newborn babies are vitamin K deficient.
- Obstructive jaundice: Since bile-containing bile salts do not reach the small intestine, fats and fat-soluble vitamins including vitamin K are not absorbed in the gut.
- Prolonged administration of broad-spectrum antibiotics destroys bacteria in the large intestine. These bacteria are an important source of vitamin K.
- Overdose of oral anticoagulants (dicoumarol)
- Dietary deficiency of vitamin K is rare.
