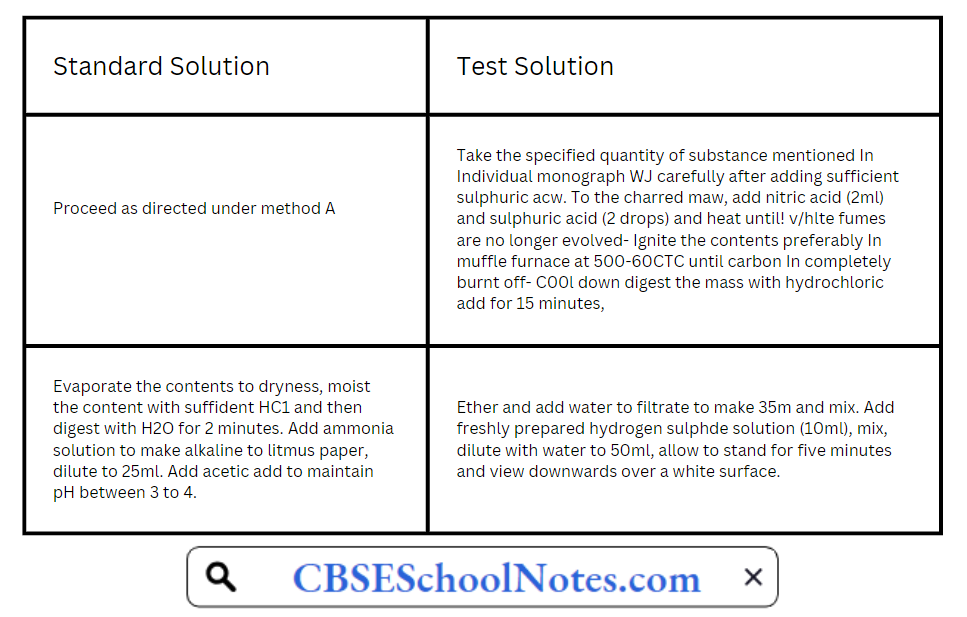
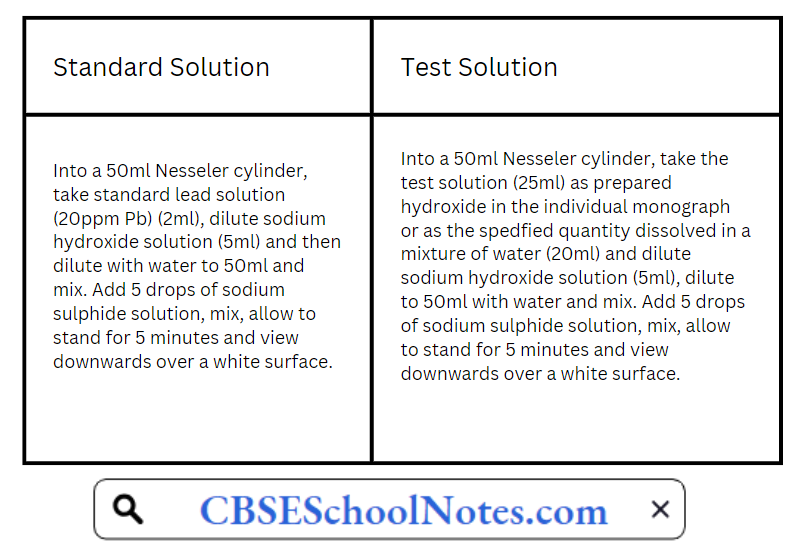
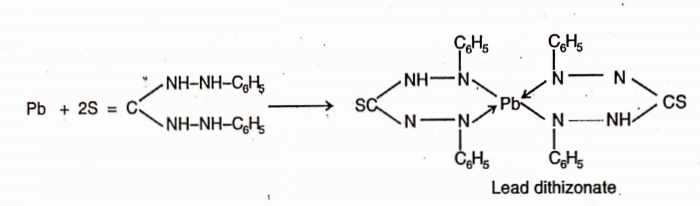
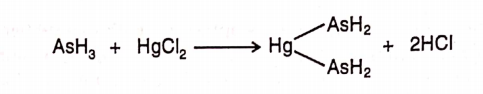
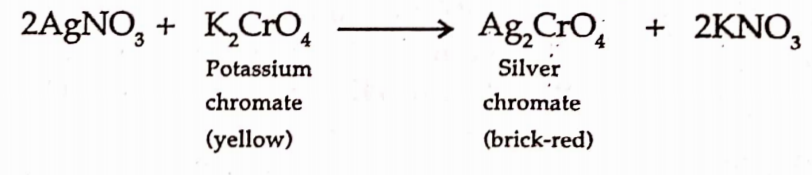
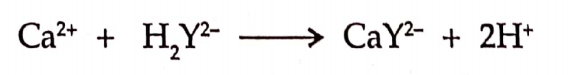
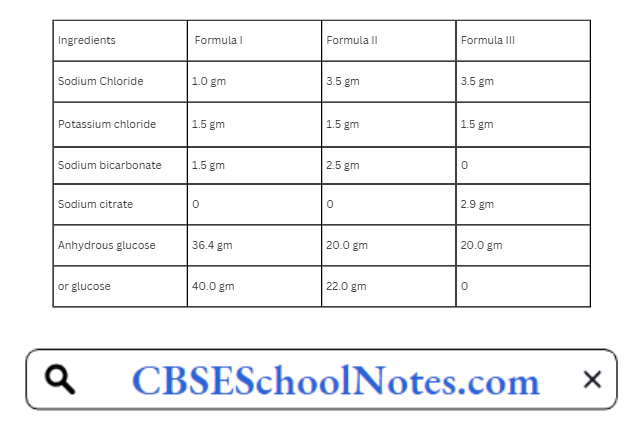
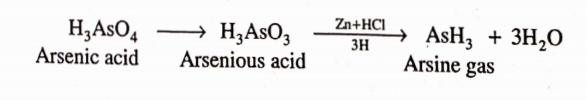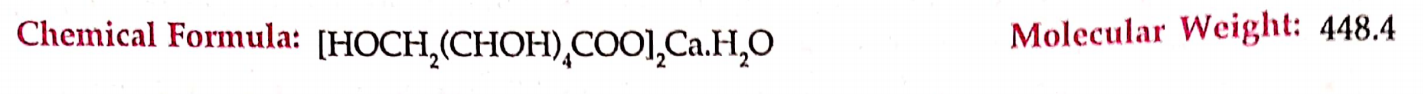CBSE Class 8 English Chapter 1 Parts of Speech
Question 1. Look at the following picture and write six to seven sentences about it. Also, discuss with your partner about each word used in the sentences in terms of parts of speech.

Answer:
The picture shows a dense forest with tall trees, lush green leaves, and sunlight filtering through the branches. A small stream flows gently between the trees, adding to the serenity of the scene. Birds are perched on the branches, chirping melodiously. The forest floor is covered with fallen leaves and wildflowers, creating a colorful carpet. The air is fresh and filled with the earthy scent of nature. This peaceful environment is home to many animals and insects. Forests like these are vital for maintaining ecological balance.
Parts of Speech:
- Forest – Noun (names a place).
- Tall – Adjective (describes the trees).
- Flows – Verb (action of the stream).
- Gently – Adverb (describes how the stream flows).
- Melodiously – Adverb (describes how the birds chirp).
- Colorful – Adjective (describes the carpet of leaves and flowers).
- Fresh – Adjective (describes the air).
Question 2. Put the words in the correct category.
everybody these Cion type skip blue Rachel our want advice throw pointed t&at smart run yourselves patience did me spell himself students Sydney excellent easy woollen wtfo modern which wHi volleyball Spanish Cdina bright uie/lern trouble skiing she be

Answer:

CBSE Class 8 English Chapter 2 The Noun
Question 1. Circle all the nouns in the following passage. Then sort them into the correct boxes.
Rehan couldn’t sleep at all that night. The little creature was afraid to close his eyes. Naveen might wake up at any time and point that awful, glaring flashlight directly at Rehan, who was crouched behind a pair of shoes in the open cabinet. Things had been likewise since Naveen began reading Do Not Enter The Monster Zoo, a collection of stories that portrays all the monsters as ruthless villains and twists the history of hatred to accuse creatures of committing things to people that people actually do to the monsters!

Answer:

Question 2. Label the objects and write their names in the correct columns, i.e., countable and uncountable.

Answer:


CBSE Class 8 English Chapter 3 Pronouns
Question 1. Look at the following pictures and describe each picture using personal pronouns in both the subjective and the objective case

Answer:

CBSE Class 8 English Chapter 4 Determiners
Question 1. Look at the picture and complete the following sentences with a word from the box. Sometimes there are two possibilities.


Question 1._________ patient (except patients with x-rays) must wait to be called.
Answer: All
Question 2. _____________patient who arrives 30 minutes late will have to wait until after the last patient has been seen.
Answer: Any
Question 3. There are blood tests __________ Wednesday and Friday.
Answer: On
Question 4. Nurse is speaking to the patients.
Answer: Each
Question 5. Flu vaccinations are free for people over 60.
Answer: All
Question 6. The boy on the left has broken_________arms.
Answer: Both
Question 7. There are ________________ blood tests on Mondays.
Answer: No
Question 8. There are three doctors, and _________________doctor is specialised in adifferent field.
Answer: Each
Question 9. ___________patients at the desk are carrying X-rays.
Answer: All
Question 10. ________________patients on the left are waiting to see a doctor.
Answer: All
Question 11. To donate blood, you can come on Fridays at___________time between 8 and 11
Answer: Any
Question 12.____________ person in the room is either a nurse or a patient.
Answer: Every
Fill in the gaps with ‘a’, ‘an’, ‘the’ or ‘O’ article.
Question 1. Where do you live?” “I live in___________flat in Gurugram. It’s _________nice flat, but___________rooms are too small. I like ___________big rooms, you know…” “I also live in____________flat It’s in Vasant Kunj
Answer: A; A;The;O;A
Question 2. When we got home,______________show had already started. We missed ___________beginning.
Answer: The; The
Question 3. Ramona has got____________new mobile phone. Her mother bought___________ mobile phone from downtown yesterday.
Answer: A; The
Question 4. My mother is____________journalist, and my father is___________engineer.
Answer: A; An
Question 5. I’ve never been to_________Zoo so i Have seen________________elephant
Answer: The; An
Question 6._______________vasco da gama Bridge across__________River Taugs is ____________ Longest Bridge in___________Europe.
Answer: The ;The ;The;noarticle
CBSE Class 8 English Chapter 5 The Present Tense
Question 1. Read the conversation and answer the questions.
Ridhima: I’m Ridhima Shetty. Are you Abhilash Singh?
Abhilash: Yes, I’m. Are you North Indian?
Ridhima: No. Vikas is North Indian and lives at Jaipur. I’m Marathi.
Abhilash: Oh, okay! I am from Delhi.
Vikas: Are you from old Delhi?
Abhilash: No, I’m from Karol Bagh, New Delhi. Is Jaipur a big city?
Vikas: Yes, it is a big city. Ridhima, are you from Mumbai?
Ridhima: Yes, I’m from Mumbai.
Abhilash: Are you and Ridhima students?
Vikas: I’m a student. She is an actress in television serials.
We are tourists in Goa.
Ridhima: Are you a student, Abhilash?
Abhilash: No, I’m not a student. I’m a doctor. I’m on holiday.
Question 1. Is Ridhima from Pune?
Answer: No, Ridhima is not from Pune; she is from Mumbai
2. Is Abhilash from Delhi?
Answer: Yes, Abhilash is from Karol Bagh, New Delhi.
3. Are Ridhima and Vikas tourists in Goa?
Answer: Yes, both Ridhima and Vikas are tourists in Goa.
4. Is Abhilash a lawyer?
Answer: No, Abhilash is not a lawyer.
5. Is Ridhima an actress?
Answer: Yes, Ridhima is an actress.
CBSE Class 8 English Chapter 6 The Past Tense
Question 1. Find and highlight 20 verbs in the given word search.

Answer:
- SING
- HAVE
- SWIM
- EAT
- DRIVE
- WALK
- READ
- TAKE
- DRINK
- WATCH
- COOK
- CLEAN
- KICK
- WASH
- LISTEN
- PLAY
- MAKE
- RIDE
- TELL
- ASK
2. Use the verbs you found in the word search to complete the sentences below in the past continuous tense
Question 1. They weren’t______________to school today; they were__________their bikes
Answer: walking, riding
Question 2. He wasn’t______________a fime; he was __________a book
Answer: riding; reading
Question 3.I was not _________________dinner at home; I was___________dinner at a restaurant
Answer: reading, eating
Question 4.She was ______________ to work; I was____________the bus
Answer: driving; taking
Question 5.We weren’t ________________tea; we were____________cakes
Answer: drinking; making
Question 6.He was not __________the house; he was __________his clothes.
Answer: cleaning ; washing
Question 7. They weren’t____________to music; they were______a song.
Answer: listening ;singing
Question 8. She wasn’t____________the drums; she was________the door
Answer: kicking ; knocking
Question 9. He wasn’t ____________in the sea; he was____________a sandcastle.
Answer: swimming;making
Question 10. I was not ________________ him to do it; I was________him.
Answer: Telling; asking
3. Fill in the sentences with the past perfect continuous tense form of the verbs given in box, and then match the sentences to the picture.


Question 1. Jim _______________ at the stars before he went to bed.
Answer: had been looking
Question 2. How long __________________ the child ________________before his dad picked him up?
Answer: had ; been crying
Question 3.Sam_____________for three hours before we me
Answer: had been sleeping
Question 4.Jatin____________a shower for half an hour before the telephone rang
Answer: had been having
Question 5. Raman said he_______________to our conversation
Answer: had not been listening
Question 6. Before the kids went outside, they____________for two hours.
Answer: had been colouring
Question 7. ___________ the littel girl ____________ the Booklet before the
Answer: Had; been painting
Question 8. ______________Jason____________ the meal for hours before the guests arrived
Answer: Had; been cooking
Question 9. Granny _________________a picture for some days she showed it bone to me.
Answer: had been painting
Question 10. Before Pete played, he _________ the net for an hour.
Answer: had been surfing
CBSE Class 8 English Chapter 7 The Future Tense
The fortune teller is predicting the future to these people. Look at the pictures and fill in the blanks with the correct form of verbs. Also, write the corresponding number of the sentence in the picture to match them appropriately.


Question: 1. Sohit__________ with a very attractive woman
Answer: Split will become
Question: 2.The Guptas _____________a new house in the countryside.
Answer: will buy
Question: 3.I have got bad news for you. Somebody_________ your car this year.
Answer: will steal
Question: 4.Bijoy___________a world record at Olympic Games.
Answer: will break
Question: 5.Somebody_________you a puppy next month
Answer: will give
Question: 6.Mr John____________a lot of money at a casino.
Answer: will lose
Question: 7. Gayatri ______________the Miss World beauty contest.
Answer: will win
Question: 8. Jessica_____________around the world.
Answer: will travel
Question: 9. Alice_________books. She’ll be very popular
Answer: will write
Question: 10. Simon ___________ the army next month.
Answer: will join
2. Write your questions for the fortune teller. Then work with a friend. One is fortune teller, the other one asks questions. Practise the dialogue and write the fortune teller’s predictions Fortune Teller Activity: Questions and Predictions
Instructions:
Partner A (Questioner): Ask the fortune teller 5-10 questions about your future.
Partner B (Fortune Teller): Answer with predictions using future tense (will/won’t + verb).
Switch roles and repeat!
Example Questions & Predictions
Fortune Teller (Predictions)
Question 1. “Will I become rich?”
Answer: “You will become rich, but not before you turn 30!”
Question 2. “Will I travel to another country next year?”
Answer: “Yes! You will travel to Japan next summer.”
Question 3. “Will I meet someone famous?”
Answer: “Hmm… you won’t meet a celebrity, but you will make a talented new friend.”
Question 4. “Will I pass my exams?”
Answer: “If you study hard, you will pass with excellent grades!”
Question 5. “Will I get a pet soon?”
Answer: “I see a small dog in your future… you will adopt one next winter.
3. Fill in the blanks using the future continuous or the future perfect tense form.

Question 1. Next week, while you are working hard at the office, I __________in the sea
Answer: will be traveling
Question 2.Mr Ghosh______________his daughter’s letter by the time his wife returns home
Answer: will have chosen
Question 3.When I arrive home, my mother ____________ on her treadmill
Answer: will be running
Question 4. When Jayanti arrives home, we _ to give her a surprise
Answer: will be ready
Question 5.Before the end of the year, Bob and his wife_ all the money they won.
Answer: will have told ; was spent
Question 6.I can’t use my computer this afternoon because a technician ____________ it
Answer: will be repairing
Question 7. Tomorrow I can’t go to the cinema. I____________my grandmother at hospital.
Answer: will be visiting
Question 8.By the end of the year, Paul_____________a job as a secretary in his
Answer: will have become
Question 9.Before next summer, John ____________ enough money to go on holiday.
Answer: will have saved
Question 10.It’s been raining for a long time. I hope the Sun ________when I get up tomorrow
Answer: will have stopped
Question 11. I am sure Karan _ in the maths test tomorrow. He will be punished by the teacher.
Answer: will be chatting
Question 12. In a few months, the police_____________dangerous criminal.
Answer: will have caught
CBSE Class 8 English Chapter 8 Models
Look at the following pictures and read the sentences given below them. Then, match the sentences which express the idea or action for each picture.


Question 1. General Ability
Answer: “He can ride a bicycle.”
Question 2. Deny Permission
Answer: “You can’t go to the party.”
Question 3. Obligation (Prohibition)
Answer: “You mustn’t smoke here. It is forbidden.”
Question 4. Possibility
Answer: “It’s very cold. It may snow.”
“If you played, you might win a lot of money.”
Question 5. Asking Permission/Request (Polite)
Answer: “May I go to the toilet?”
Question 6. Absence of Obligation
Answer: “It’s Sunday. He doesn’t have to get up early.”
Answer: “You needn’t pay for the pizza. It’s free.”
Question 7.Advice
Answer: “You should brush your teeth after every meal.”
“You shouldn’t smoke. It’s bad for your health.”
“The lights went out. You ought to light a candle.”
Question 8. Request
Answer: “It is raining. Will you lend me an umbrella?”
Question 9. Past Habit
Answer: “I would go to the beach when I was a little girl.”
Question 10. Obligation (Necessity)
Answer: “You must do your homework.”
“He has a toothache. He has to go to the dentist.”
Question 11. Offer
Answer: “Shall I help you carry your luggage?”
Question 12.Certainty
Answer: (No direct example; implied in sentences like “It must be true.”)
2. Perform this activity with your partner. Read the following sentences with a blank and give options to your partner. Your partner will have to choose the correct option
Question 1. If you’re in a temple, you____________speak loudly
- have to
- mustn’t
- should
Answer: mustn’t
Question 2. Don’t forget to take an umbrella. It________ rain letter
- might
- must
- should
Answer: might
Question 3._______________ I wear jewellery to school?
- Should
- Shall
- May
Answer: May
Question 4. Shalini ___________ be ill. I’ve just seen her playing
- can’t
- can
- has to
Answer: can’t
Question 5. We________chew Gum in class.
- should
- must’take
Answer: mustn’t
Question 6.You ______________be hungry. You’ve just eaten lunch
- can’t
- might
- can
Answer: can’t
Question 7. I’ve just bought a lottery ticket. I____________become rich soon
- have to
- need to
- must
Answer: must
Question 8. Mr Gautam_________be away this weekend, but we’re not sure
- may
- might
- ought to
Answer: might
Question 9. Rajeev has finally got the job. He____________ be really pleased.
- must
- could
- can
Answer: must
Question 10. Mom____________do the shopping. Dad has already done it
- has to
- needs to
- doesn’t need to
Answer: doesn’t need to
Question 11. Niti couldn’t find her car keys. They_____________be in her bag
- may
- could
- couldn’t
Answer: could
Question 12. Piyush __________________ be at home. His car is outside
- may
- mustn’t
- must
Answer: must
Question 13. You__________vacuum the carpet. Raju already cleaned the room
- can’t
- should
- don’t have to
Answer: don’t have to
Question 14. He_______________ speak Spanish fluently. He’s been there for only a week.
- can
- can’t
- mustn’t
Answer: can’t
Question 15. Whose pen is it? I’m not sure. It__________be Sandra’s
- might
- can
- must
Answer: might
Question 16. Where can I find Vyom? ‘He_____________be in the garden.
- ought to
- may
- must
Answer: must
CBSE Class 8 English Chapter 9 Verbs – Non-Finites
1. Choose and circle the correct participle form in each sentence given below. Also, match the description with the picture.

Question 1. Sunidhi did a lot of housework yesterday. She was really tired/tiring when she finished.
Answer: Tired
Question 2. In my dream last night, I saw a frightened/ frightening monster.
Answer: frightened
Question 3. Sushmita felt very excited/exciting when her v parents took her to the amusement park.
Answer: Excited
Question 4. Mr Parekh gets bored/boring after driving in a traffic jam for a long time.
Answer: bored
Question 5. Being a mechanic is a tired/tiring job. You must be strong and patient to work as a mechanic.
Answer: Tiring
Question 6. John looked exhausted/exhausting after he spent some time in the gym.
Answer: exhausted
Question 7. Mr Jacob seemed shocked/shocking when he heard that his wife had an accident.
Answer: shocked
Question 8. The film was really excited/exciting, so Ritu and Jai watched it twice.
Answer: exciting
Question 9. Mr Dev is feeling relaxed/relaxing after a lot of work.
Answer: relaxed
Question 10. Nobody wants to listen to Dushyant because ‘ he is a bored /boring man
Answer: boring
CBSE Class 8 English Chapter 10 Active and Passive Voice
Neelima is a Bengaluru-based student who lives in a Park Street apartment. When she returned home from college in the afternoon, she discovered that her room had been broken into.
You were one of the officers that responded to her plea for assistance. Examine the image and create a composition describing what occurred in the room.
Remember to use the sentences both in active and passive voice. Then, ask your partner to categorise the sentences both in active and passive voice separately.

Active Voice (Subject performs the action):
- The thief smashed the window to enter Neelima’s room.
- He scattered her books and papers across the floor.
- Neelima reported the incident to the police immediately.
- The officers collected fingerprints from the broken drawer.
Passive Voice (Subject receives the action):
- The window was smashed by the thief to gain entry.
- Neelima’s books and papers were scattered across the floor.
- The incident was reported to the police by Neelima.
- Fingerprints were collected from the broken drawer by the officers.
Task for Partner:
Categorize these sentences into Active and Passive Voice:
- The thief stole Neelima’s laptop. (Active)
- The jewelry was hidden under the bed by Neelima. (Passive)
- The police questioned the neighbors. (Active)
- A suspicious footprint was found near the window. (Passive)
CBSE Class 8 English Chapter 11 Adjectives- Degrees of Comparison
Read the following story and fill in the blanks with the correct form of adjectives given below.

Question 1. Once upon a time, two brothers named Ed and Ted always competed against each other. No matter how hard Ted tried, Ed always did everything__________________
Answer: Better
Question 2. Ed was one year_________his brother Ted. And when they measured their heights, Ed was always one foot ___________ his brother.
Answer: older, taller
Question 3. They loved kicking the soccer ball, but Ed always kicked the ball_____________ his brother Ted no matter how hard Ted tried. When they played basketball too, Ed always jumped________________ his brother too. Finally, Ed was always a couple of seconds _____________Ted when they ran to the finish line
Answer: farther,higher, faster
Question 4. Ted was going to give up on being the__________because his brother was always_________________ than everything he did.
Answer: best, better
Question 5. Ed and Ted were both exhausted. Their mom called them back into the house for dinner. They sat down at the table, but Ted was__________________ his brother because he ate his whole meal.
Answer: better
Question 6. At that point, both boys realised that you couldn’t be ____________at everything than your brother in life
Answer: better
2. Look at the following pictures and describe each picture using the adjectives given below them. You can use any degree of adjective.

Question 1. Strong
Answer: The weightlifter is strong, but his coach is stronger, and the champion is the strongest of all.
Question 2. Frightened
Answer: The child looks frightened, the dog seems more frightened, but the cat is the most frightened by the thunderstorm.
Question 3. Poor
Answer: The beggar is poor, the homeless man is poorer, but the orphan in the war-torn country is the poorest.
Question 4. Desperate
Answer: The man searching for water appears desperate, the lost hiker looks more desperate, but the trapped miner is the most desperate.
Question 5. Happy
Answer: The baby is happy, the couple is happier, and the grandparents are the happiest at the family reunion.
Question 6. Hungry
Answer: The student is hungry, the marathon runner is hungrier, but the starving child is the hungriest.
Question 7. Proud
Answer: The artist is proud, the scientist is prouder, and the soldier receiving a medal is the proudest.
Question 8. Fat
Answer: The house cat is fat, the raccoon is fatter, but the bear is the fattest of them all.
Question 9. Beautiful
Answer: The garden is beautiful, the sunset is more beautiful, but the mountain view is the most beautiful.
Question 10. Exhausted
Answer: The worker is exhausted, the nurse is more exhausted, but the firefighter after a 24-hour shift is the most exhausted.
Question 11. Doubtful
Answer: The student is doubtful, the investor is more doubtful, but the scientist questioning a theory is the most doubtful.
Question 12. Ill
Answer: The child is ill, the elderly woman is more ill, but the patient in the ICU is the most ill.
Question 13. Cold
Answer: The morning is cold, the winter night is colder, but the Arctic storm is the coldest.
Question 14. Healthy
Answer: The apple is healthy, the salad is healthier, but the homegrown organic meal is the healthiest.
Question 15. Funny
Answer: The clown is funny, the comedian is funnier, but the grandfather’s jokes are the funniest.
Question 16. Religious
Answer: The monk is religious, the priest is more religious, but the hermit is the most religious.
Question 17. Eager
Answer: The puppy is eager, the child on Christmas morning is more eager, but the athlete before the Olympics is the most eager.
Question 18. Generous
Answer: The neighbor is generous, the philanthropist is more generous, but the anonymous donor is the most generous.
Question 19. Delicious
Answer: The bread is delicious, the cake is more delicious, but Grandma’s homemade pie is the most delicious.
Question 20. Lazy
Answer: The cat is lazy, the sloth is lazier, but the teenager on summer vacation is the laziest.
CBSE Class 8 English Chapter 12 Adverbs
1. Make two teams and perform this activity. One student from each team will have to come to the board to write a story. As she/he writes, he must use several adverbs and adjectives to make the story most interesting.
The students from each team will recognise adverbs and adjectives in the opposite team’s story. Write each word you add in the adverb or adjective list based on how it is used in the text. The team whose members identify the adverbs and adjectives more correctly, will win.
Answer :
Adverb/Adjective Story Challenge
Objective: Teams compete to write stories packed with adverbs/adjectives, then identify them in the opponent’s story. The most accurate team wins!
Setup (5 minutes)
1. Divide the class into Team A and Team B.
2. Assign roles:
1 Writer per team (writes the story on the board).
3-4 Identifiers (spot adverbs/adjectives in the opponent’s story).
1 Judge (teacher or selected student verifies answers).
Rules & Gameplay
1. Story Writing (5 minutes/team):
Each team writes a 5-sentence story on the board.
Must include:
5+ adverbs (e.g., suddenly, loudly, carefully).
5+ adjectives (e.g., spooky, golden, exhausted).
Identification Round (3 minutes/team):
Opposite team underlines adverbs/adjectives in the rival’s story.
1 point per correct guess; -1 for incorrect guesses.
Winning:
2. Read the following sentences. If the highlighted word is an adjective, write ADJ, and if it is an adverb, write ADV.
Question 1. The cheese on this cracker tastes strange.___________________
Answer: ADJ
Question 2. I go to the gym regularly.___________________________
Answer: ADV
Question 3. She gazed at me with empty eyes. __________________
Answer: ADJ
Question 4. Have you eaten an apple lately? _______________________
Answer: ADV
Question 5. The dog howled wildly in its cage.__________________________
Answer: ADV
Question 6. I think that both sides are even now. ___________________________
Answer: ADJ
Question 7. The instructor arrived promptly at eight. ______________
Answer: ADV
Question 8. Our regular office hours are Monday to Friday from 9 am to 5 pm._______________
Answer: ADJ
Question 9. The teams were matched evenly._________________
Answer: ADV
Question 10. The homemade food was quite tasty__________________
Answer: ADV
CBSE Class 8 English Chapter 13 Adverbs
Question 1. Work in pairs and perform the activity. Ask your partner the following things given in the table and complete it.
Answer:

CBSE Class 8 English Chapter 14 Phrasal Verbs
1. Perform this activity in pairs. One student will read out the phrasal verb and its meaning, while the other student will read the sentence. Then, both the students will write different sentences using the same phrasal verb in their notebook.

Answer:

Phrasal Verbs Pair Activity Guide
Objective: Practice phrasal verbs through collaborative sentence-building.
Setup:
Pair students (Student A and Student B).
Materials: Notebooks, printed phrasal verb table (or displayed on screen).
Step-by-Step Instructions:
1. Role Assignment (2 minutes)
Student A: Reads the phrasal verb + meaning aloud.
Example: “Put on – to place something on the stove.”
Student B: Reads the example sentence aloud.
Example: “I’ll put the kettle on for tea.
Sentence Creation (5 minutes/pair)
Both students write 2 new sentences in their notebooks using the same phrasal verb.
Example for “put on”:
“She put the soup on to simmer.”
“Don’t forget to put on your apron before cooking!
2. Look at the following words which go along with the word ‘get’ to make the phrasal verbs. Understand their meanings and fill in the blanks with suitable words.

Question 1. My brother is an early bird. He gets __________at 5 am every day.
Answer: up
Question 2. How can Sam get__________on his salary?
Answer: by
Question 3. Parul usually fails to get her jokes______________
Answer: across
Question 4. Nimit got__________his exams without any effort
Answer: through
Question 5. I’ve never got _____________with my brother-in-law.
Answer: along
Question 6.If this secret gets________you’ll be in trouble
Answer: out
Question 7. We’ve climbed the fence easily, but now we can’t get_____________
Answer: down
Question 8. You should get_____________with your homework right now
Answer: on
Question 9. The burglar got___________before we came back home.
Answer: away
Question 10. When Moly got__________________the plane, she saw all her family waiting for her.
Answer: off
Question 11. Rishabh__________________has beenThanksunemployedto his forparents’six months.help! He is getting
Answer: by
Question 12. After the concert the audience got ________and applauded for ten minuts.
Answer: up
Question 13. Can you tell me where I should get_____________?
Answer: off
Question 14. How did Meenu get____________here? She hasn’t got the key, has she?
Answer: in
Question 15. It took me two weeks to get____________the terrible flu.
Answer: over
CBSE Class 8 English Chapter 15 Structure of Sentences
1. In each of the following sentences, underline the subject once and the predicate twice.
Question 1. Fables such as “Little Red Riding Hood” have given wolves a bad reputation.
Answer: Fables such as “Little Red Riding Hood” have given wolves a bad reputation.
Subject: Fables such as “Little Red Riding Hood”
Predicate: have given wolves a bad reputation
Question 2. Ranchers in areas with wolves dislike the animals for attacking their livestock.
Answer: Ranchers in areas with wolves dislike the animals for attacking their livestock.
Subject: Ranchers in areas with wolves
Predicate: dislike the animals for attacking their livestock
Question 3. In the city lived many poor people.
Answer: In the city lived many poor people.
Subject: many poor people (Note: This is an inverted sentence; the subject follows the verb.)
Predicate: In the city lived
Question 4. Out of the woods came a bear.
Answer: Out of the woods came a bear.
Subject: a bear (Inverted structure; subject follows the verb.)
Predicate: Out of the woods came
Question 5. Just around the corner was the parade.
Answer: Just around the corner was the parade.
Subject: the parade (Inverted structure.)
Predicate: Just around the corner was
Question 6. In the fall, the leaves cover the ground.
Answer: In the fall, the leaves cover the ground.
Subject: the leaves
Predicate: cover the ground
Question 7. Their sudden appearance quickly aroused the townspeople’s curiosity.
Answer: Their sudden appearance quickly aroused the townspeople’s curiosity.
Subject: Their sudden appearance
Predicate: quickly aroused the townspeople’s curiosity
Question 8. With keen interest, the townspeople observed the prospectors’ movements.
Answer: With keen interest, the townspeople observed the prospectors’ movements.
Subject: the townspeople
Predicate: observed the prospectors’ movements
Question 9. Nobody in the town knew anything about the strangers.
Answer: Nobody in the town knew anything about the strangers.
Subject: Nobody in the town
Predicate: knew anything about the strangers
Question 10. The children’s books and backpacks were stolen while they were at lunch.
Answer: he children’s books and backpacks were stolen while they were at lunch.
Subject: The children’s books and backpacks
Predicate: were stolen while they were at lunch
Question 11. The zoo and the ice cream store are my favourite destinations.
Answer: The zoo and the ice cream store are my favourite destinations.
Subject: The zoo and the ice cream store
Predicate: are my favourite destinations
Question 12. The teacher and her assistant told the student and his parents that there would be a quiz in the next week.
Answer: The teacher and her assistant told the student and his parents that there would be a quiz in the next week.
Subject: The teacher and her assistant
Predicate: told the student and his parents that there would be a quiz in the next week
B. Work in pairs. One student will rewrite the following sentences by changing the position of direct and indirect objects. Then his/her partner will replace the indirect objects with personal pronouns.
Question 1. Grandma will cook my friends a delicious meal. Grandma will cook my friends a delicious meal.
Swapped: Grandma will cook a delicious meal for my friends.
Pronoun: Grandma will cook a delicious meal for them.
Question 2. Did Ravi bring Rahul the new book? Did Ravi bring Rahul the new book?
Swapped: Did Ravi bring the new book to Rahul?
Pronoun: Did Ravi bring the new book to him?
Question 3. Mom left Sunita with two oranges. Mom left Sunita two oranges.
Swapped: Mom left two oranges for Sunita.
Pronoun: Mom left two oranges for her.
Question 4. The teacher told the students funny stories.
The teacher told the students funny stories.
Swapped: The teacher told funny stories to the students.
Pronoun: The teacher told funny stories to them.
Question 5. Who will lend my brother some money?
Who will lend my brother some money?
Swapped: Who will lend some money to my brother?
Pronoun: Who will lend some money to him?
Question 6. Gagan gave Tarun a ride to the station. Gagan gave Tarun a ride to the station.
Swapped: Gagan gave a ride to the station to Tarun.
Pronoun: Gagan gave a ride to the station to him.
Question 7. The teacher gave the class a new project.
The teacher gave the class a new project.
Swapped: The teacher gave a new project to the class.
Pronoun: The teacher gave a new project to them.
Question 8. My mom bought my brother a new dress.
My mom bought my brother a new dress.
Swapped: My mom bought a new dress for my brother.
Pronoun: My mom bought a new dress for him.
Question 9. They bought my parents some CDs.
They bought my parents some CDs.
Swapped: They bought some CDs for my parents.
Pronoun: They bought some CDs for them.
Question 10. Tanuj showed Ms Sharma his project.
Tanuj showed Ms Sharma his project.
Swapped: Tanuj showed his project to Ms Sharma.
Pronoun: Tanuj showed his project to her.
CBSE Class 8 English Chapter 16 The Phrase and the Clause
1. Fill in the blanks with appropriate phrases
Question 1. The gift_________________________is from me.
Answer: wrapped in blue paper
Question 2.That song_______________reminds me of you
Answer: playing on the radio
Question 3. She loved him because he was a man_______________
Answer: of integrity
Question 4. Students_______________usually do well.
Answer: who study regularly
Question 5. He denied________________
Answer: stealing the cookies
Question 6. The wicked man loves__________________
Answer: causing trouble
Question 7. The poor man wanted________________________
Answer: to feed his family
Question 8. Horses prefer______________________
Answer: running in open fields
Question 9. People____________________are extremely rude
Answer: who interrupt others
Question 10. That desk______________looks nice in the dining room.
Answer: with the carved legs
Question 11. Have you ever tried__________________?
Answer: making homemade pasta
Question 12._______________________ surprises me.
Answer: Her sudden success
CBSE Class 8 English Chapter 17 Sentences – Simple, Compound, Complex
1. Read the following instructions and tick (/) the correct option
Question 1. The simple sentences in a compound sentence can be joined together with a comma and a__________________
- noun
- preposition
- conjunction
- verb
Answer:(3) conjunction
Question 2. The simple sentences in a compound sentence can be joined together with a _______________
- period
- semicolon
- question mark
- exclamation mark
Answer:(2) semicolon
Question 3. The simple sentences in compound sentences are also known as independent __________________
- phrases
- clauses
- words
- predicates
Answer: (2) clauses
Question 4. Choose the appropriate coordinating conjunction to join these simple sentences into a compound sentence.
Nitin ordered a burger,__________________Manu chose a sandwich
- if
- nor
- and
- or
Answer:(3) and
Question 5. Choose the subordinate clause in the given complex sentence When I went to the carnival, I rode on the Ferris Wheel.
- When I went to the carnival
- I went to the carnival
- I rode
- I rode on the Ferris Wheel
Answer: (1) When I went to the carnival
Question 6. Choose the subordinate clause in the given complex sentence. Varsha wrote down my e-mail address, so she would not forget it.
- Varsha wrote down my e-mail address
- my e-mail address
- so she would not forget it
- she would not
Answer:(3) so she would not forget it
Question 7. A complex sentence has one independent clause and one or more subordinate clauses.
- True
- False
Answer: (1) True
Question 8. Choose the subordinate clause in the given complex sentence. Alexander, who is fourteen years old, loves to skateboard.
- Alexander
- who is fourteen years old
- Alexander loves to skateboard
- loves to skateboard
Answer: (2) who is fourteen years old
CBSE Class 8 English Chapter 18 Noun Clauses
1. Choose the most appropriate noun phrase.
1. I was showing the tourist____________
- when to go home
- what to sell at the market
- How to get to the hotel
Answer: 3. how to get to the hotel
2. Mr Hari told the driver__________
- where to send him
- what to think
- what to do at the meeting
Answer: 1. where to send him
3. One of the players forgot
- when to turn up for practice
- how to run away
- where to buy shoes from
Answer: 1. when to turn up for practice
4. The chef is wondering ____________
- when to brush his teeth
- what to prepare for the important dinner
- how to wash the car
Answer: 2. what to prepare for the important dinner
5. The librarian told Peter________________
- when to start work
- how to lock up the place
- what to look out for on the road
Answer: 2. how to lock up the place
6. At the party, she showed him _________
- what to grow
- how to dance
- when to sleep
Answer: 2. how to dance
7. The sergeant is showing the soldier_____________
- what to sing at the concert
- when to grow his beard
- how to hold the rifle
Answer: 3. how to hold the rifle
8. The usher showed us___________
- how to take photographs
- where to sit
- when to comb our hair
Answer: 2. where to sit
9. Our teacher told us _______________
- what to do with the test paper
- how to lock up the place
- where to look for the survivors
Answer: 1. what to do with the test paper
10. The stewardess shows the passengers_________________
- when to start work
- where to land the plane
- how to fasten the seat belt
Answer: 3. how to fasten the seat belt
CBSE Class 8 English Chapter 19 Adverb Clauses
Fill In the Blanks With Appropriate Adverbs
1. He joined the army ____________
Answer: after he finished school
2. He doesn’t like anyone talking to him _____________
Answer: while he’s watching TV
3. By the time we got back ___________
Answer: the children were asleep
4. She turned off the lights_____________
Answer: before she left the house
5. They acted_______________
Answer: as if they were very excited
6. You can’t leave the room________________
Answer: until the lesson finishes
7. They bring us flowers__________________
Answer: whenever they come for a visit
8. Wherever Julie goes,___________________
Answer: she takes her daughter
CBSE Class 8 English Chapter 20 Short Compositions
1. Look at the following picture and write a factual description.

Answer:
Factual Description of a Supermarket
The image depicts a large, well-lit supermarket interior with organized aisles and numerous products on display. The store features bright fluorescent lighting and clean, polished floors. Shelves are neatly stacked with packaged goods, including canned foods, cereals, and snacks, arranged in rows for easy access.
In the foreground, shopping carts and baskets are available for customers near the entrance. Several shoppers can be seen browsing the aisles, some pushing carts while others carry baskets. Staff members in uniform are visible, with one restocking shelves and another operating a cash register at the checkout counter.
The supermarket has clear signage above each aisle, indicating product categories such as “Dairy,” “Bakery,” and “Frozen Foods.” Refrigerated sections line the sides of the store, displaying fresh produce, dairy items, and chilled beverages. The checkout area includes multiple lanes with conveyor belts, digital scanners, and payment terminals.
Promotional banners and price tags are prominently displayed throughout the store, highlighting discounts and special offers. The overall atmosphere appears busy yet orderly, with a focus on convenience and efficiency for customers.
CBSE Class 8 English Chapter 21 Comprehension Passages
Read the following text and answer the questions that follow.
When another old cave is discovered in the south of France, it is not usually news. Rather, it is an ordinary event. Such discoveries are so frequent these days that hardly anybody pays heed to them.
However, when the Lascaux cave complex was discovered in 1940, the world was amazed. Painted directly on its walls were hundreds of scenes showing how people lived thousands of years ago.
The scenes show people hunting animals, such as bison or wild cats. Other images depict birds and, most noticeably, horses, which appear in more than
300 wall images, by far outnumbering all other animals. Early artist drawing these animals accomplished a monumental and difficult task. They did not limit themselves to the easily accessible walls but carried their painting materials to spaces that required climbing steep walls or crawling into narrow passages in the Lascaux complex.
Unfortunately, the paintings have been exposed to the destructive action of water and temperature changes, which easily wear the images away. Because the Lascaux caves have many entrances, air movement has also damaged the images inside.
Although they are not out in the open air, where natural light would have destroyed them long ago, many of the images have deteriorated and are barely recognizable. To prevent further damage, the site was closed to tourists in 1963, 23 years after it was discovered.
Question 1. Which title best summarizes the main idea of the passage?
- Wild Animals in Art
- Hidden Prehistoric Paintings
- Exploring Caves Respectfully
- Determining the Age of French Caves
Answer: (2) Hidden Prehistoric Paintings
Question 2. In line 4, the phrase ‘pays heed to’ is closest in meaning to_______
- discovers
- watches
- notices
- buys
Answer: (3) notices
Question 3. Based on the passage, what is the significance of the south of France?
Answer: Based on the passage, the significance of the south of France lies in its rich archaeological and artistic heritage, particularly as the location where the Lascaux cave complex was discovered.
Question 4. Early artists did not limit themselves to the easily accessible walls but carried their painting materials to spaces that required climbing steep walls. What does this indicate about them?
Answer: It indicates that the early artists were determined and skilled.
Question 5. Other images depict birds and, most noticeably, horses, which appear in more than 300 wall images, by far outnumbering all other animals. Choose another word for ‘depict’ from the words given below.
- show
- hunt
- count
- draw
Answer: (1) show
Question 6. Why was painting inside the Lascaux complex a difficult task?
- It was completely dark inside.
- The caves were full of wild animals.
- Painting materials were hard to find.
- Many painting spaces were difficult to reach
Answer: (4) Many painting spaces were difficult to reach.
Question 7. According to the passage, all of the following have caused damage to the paintings except____________
- temperature changes
- air movement
- water
- light
Answer: (4) light
Question 8. Would you like to explore new caves? Explain your opinion giving four reasons from the text.
Answer:
Opinion with reasons from the text:
Yes, I would explore new caves because:
- They may contain historical art (like Lascaux’s prehistoric paintings).
- Discoveries can reveal ancient human lifestyles (hunting scenes).
- Caves challenge explorers with hard-to-reach spaces (steep/narrow areas).
- They are time-sensitive (paintings deteriorate, so documenting them is urgent).
No, because:
- Fragility: Human presence can damage art (as in Lascaux’s closure).
- Danger: Climbing/crawling poses risks (like the artists faced).
- Preservation: Some caves should remain undisturbed (to prevent further damage).
- Limited access: Many caves are restricted (e.g., Lascaux closed in 1963).