Haematinics are substances required in the formation of blood in process of hematopoiesis and are used for treatment of anaemias. The main hematinics are B12 & folate.
These drugs increase the number of red blood cells and the amount of hemoglobin to normal level and above when they are below normal.
Anaemia occurs when the balance between production and reduction of red blood cells is disturbed
- Due to blood loss
- Impaired red cell formation due to
- Deficiency of essential factors: Iron, folic acid vitamin B12.
- Bone marrow depression, erythropoietin deficiency
- Increased destruction of RBCs (Haemolyticanaemia)
Ferrous Sulphate
Synonyms: Green vitriol
Molecular formula: FeSO4.7H2O
Molecular weight: 278
It contains not less than 98% and not more than 105% of FeSO4.7H2O
Preparation
When iron is treated with dilute H2SO4, iron dissolve and form ferrous sulphate and hydrogen gas is liberated.
Read and Learn More Pharmaceutical Inorganic Chemistry Notes

Ferrous sulphate Is obtained commercially by exposing the moist Iron pyrites to air when slow oxidation takes place.
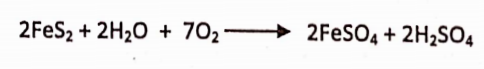
Properties
Physical properties:
It occurs as transparent, pale bluish green crystalline powder; odourless; metallic in soluble in boiling water, freely soluble in water but practically insoluble in alcohol.
Chemical properties:
When it is treated with ceric ammonium sulphate [(NH4)4Ce(SO4)42 H20] in acidic medium, it reduces ceric ion.

Assay:
Ferrous sulphate solution may bring about discolouration of the teeth in contract. The assay is based on oxidation-reduction (redox) titration. An acidified solution of substance is titrated with ceric.
Ammonium sulphate in presence of sulphuric acid using ferrous sulphate solution as an indicator. Weigh accurately 1 gm of ferrous sulphate (FeSO4) and dissolve in 20 ml of diluted sulphuric acid in 30ml. of water. Titrate the contents of the flask with 0.1 N potassium permanganate (KMnO4) until a permanent pink colour is obtained.
Uses:
It is used as hematinic in the treatment of iron deficiency anaemia. It is also used to dye fabrics and in tanning leather.
- White nitric oxide, ferrous sulphate forms black colourednitroso ferrous sulphate FeSO2.NO.
- On heating it decomposes into ferric oxide sulphur dioxide &nelpherotrixodide.
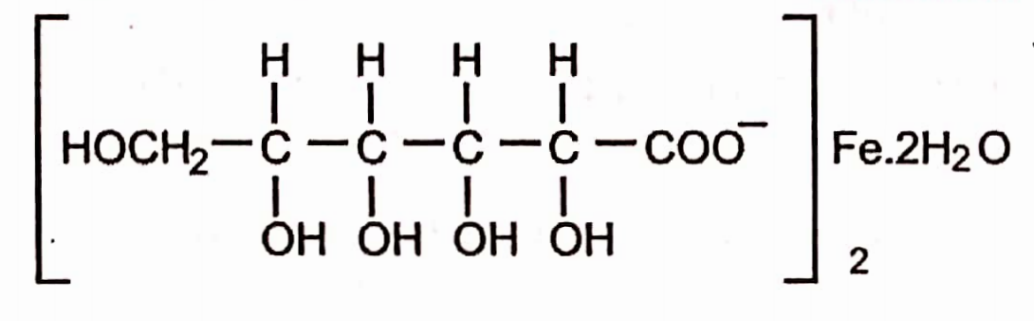
Ferrous Gluconate
Molecular formula:-C12H22FeO14.2H2O
Molecular weight: 482.2
Preparation
1 . It is obtained by dissolving ferrous carbonate in calculated amount of gluconic acid. First of all gluconic acid is prepared by the oxidation of glucose.
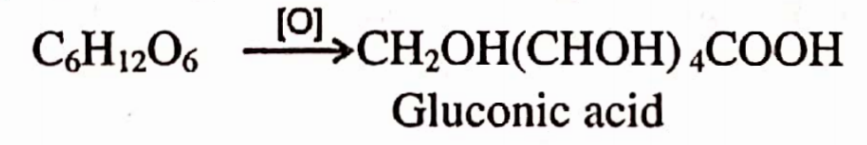
Gluconic acid is treated with BaCl2 solution which is then treated with FeS04 solution. BaS04 precipitates out & is removed by filtration.
Properties
- It occurs as a fine, yellowish gray & pale greenish yellow powder or granules with slight burnt sugar.
- It is freely but slowly soluble in water.
Assay
The assay is based on the oxidation-reduction titration. To a weighed amount (l,5gm) dissolved in water and 2N sulphuric acid, zinc powder is added & kept for 20 minutes, filtered and the filter is washed with the water.
To the combined liberate and washing ferrous sulphate solution is added & titrated with 0.1N ceric ammonium sulphate until the colour is change from orange to green. A blank determination is performed.
Use
1 . It is used for the preparation of ferrous Gluconate tablets.
Haematinics Very Short Answer Questions
Question. 1. What are haematinics?
Answer. Haematinics are substances required in the formation of blood. These drugs are used for the treatment of anaemia. These drugs increase the number of red blood cells and
the amount of haemoglobin is raised to normal level and above when they are below normal.
Haematinics Fill In The Blanks
1. Total body iron in an adult person is…………..
Answer:2.5 gm to 5 gms
2. Vitamin K] is also known as………….. is used in prevention of haemorrhagic disease of new borne.
Answer: Phytonadione
