Antimicrobials
An antimicrobial (disinfectant and antiseptic) is a substance that kills or inhibits the growth of microorganisms such as bacteria, fungi or protozoans ntimicrobial drugs either kill microbes (microbiocidial) or prevent the growth of microbes (microbiostatic).
Specific terminology describes exact mode or mechanism of action.
Antiseptics: Antiseptic is a substance that prevents or arrests the growth of action of microorganisms. It acts by inhibiting their activity or destroying them especially of agents applied to living tissue like surface of skin, application of antiseptic dressings on wounds.
The drugs like phenol, iodine, boric acid, cetrimide are the examples of antiseptics.
Disinfectants: These are the drugs or substances used either to kill bacteria or prevent their growth or multiplication. It is used on non-living objects or outside the body.
For example : Disinfection of surgical instruments, sputum and urine containers on floor. Commonly used as disinfectants includes cresol, phenol.
Germicides: These are the substances which kill microorganisms. They act by oxidation of bacterial protoplasm, by denaturation of bacterial enzymes and proteins, by increasing permeability of bacterial cell membrane.
Potency of germicide is expressed by Phenol Coefficient. Specific terminology like bacteriocide (against bacteria), fungicide (against fungi), virucide (against virus) etc. denotes exact action.
Bacteriostatics: These are the substances which primarily function by inhibiting the growth of bacteria. Thus, bacteriostatic drugs or agents do not kill but arrest the growth of bacteria.
Read and Learn More Pharmaceutical Inorganic Chemistry Notes
Sanitizers: Sanitizer is the process of rendering sanitary by reducing the number of bacterial contaminants. These are used to maintain general public health standards.
Santization can be achieved only with surfaces and articles that are physically clean in addition to possess low bacterial counts or that are free from most vegetative microbes.
High cone, of sanitizers also cause local cellular damage. Inorganic compounds generally exhibit antimicrobial action by involving either of the following three mechanisms :
- Oxidation
- Halogenation
- Protein precipitation.
Boric Acid
Chemical Formula: H3BO3
Molecular Weight: 61.83
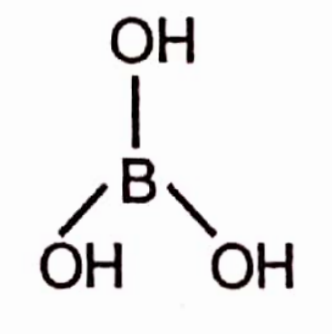
Synonyms : Hydrogen borate, boracic acid, orthoboric acid, acidum boricum.
It is widely distributed in sea water, plants and fruits. It is also available in the combined form as its largest natural source.
Method of Preparation
It is prepared by reading hydrochloric or sulphuric acid with the native borax.
The solution is filtered. The crystals obtained are washed and then allowed to dry at room temperature.
Properties
- It occurs as colourless or white crystals.
- It is slightly soluble in water and in alcohol.
- It is odourless with slightly acidic and bitter taste.
- On heating, it decomposes to form metaboric acid HBOr
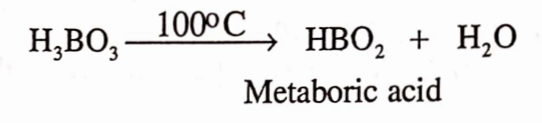
Uses: Boric acid can be used as an antiseptic for minor bums or cuts. It is used in dressings or salves. It is applied in a very dilute solution as an eye wash. As an antibacterial or antimicrobial compound, it can also be used as an acne treatment.
Borax
Chemical Formula: Na2B4O7.10H2O
Molecular Weight: 381.4
Synonyms
Sodium Borate, Sodium Tetraborate, Sodium pyroborate
Properties
It occurs as colourless crystalline or white crystalline powder. It is having saline and alkaline taste. It loses all its water of crystallisation on ignition. It is odourless.
It efflorescences in dry air. It reacts with zinc salts to give zinc borate. Acidified and aqueous solution of sodium tetraborate on heating yield crystals of botfc acid.
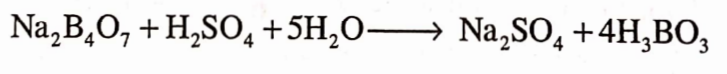
Uses
It finds use externally for eye washes and also used as an emulsifying agent for certain oils.
It is also used for softening of water.
Hydrogen Peroxide
Chemical Formula: H2O2
Molecular Weight: 34.016
It is an aqueous solution of hydrogen peroxide. It is having not less than 6% w/w of H O. which corresponds to about 20 times its volume of available 2: oxygen.
It was discovered by French chemist Thenard.
Methods of Preparation
In laboratory, it is prepared by Merck’s process. It is prepared by adding calculated V amounts of sodium peroxide to ice cold dilute (20%) solution of H2SO4.
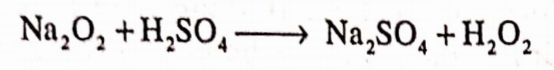
It is also prepared by the action of sulphuric acid or phosphoric acid on hydrated barium peroxide Ba02.8H20
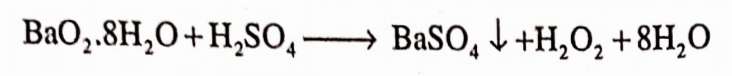
On commercial scale, H202 can be prepared by the electrolysis of 50% H2S04 solution. In a cell, peroxy sulphuric acid is formed at the anode.
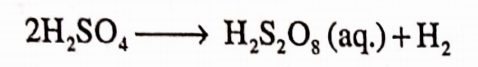
It is also obtained by decomposing barium peroxide with phosphoric acid or by passing carbon dioxide through a solution of barium peroxide in water.
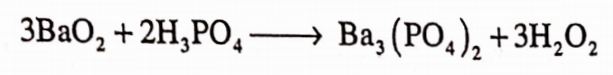
Properties
Hydrogen peroxide is clear, colourless syrupy liquid. It is odourless or may have an odour resembling to that of ozone unstable liquid. It has a specfic gravity of about 1.463 at 0°C. It is a strong oxidizing agent. It is soluble with water from which it can be extracted with solvent ether. It rapidly decomposes in contact with oxidizable organic matter and with metals.
Storage
H2O2 is not stored in glass bottles since the alkali metal oxides present in glass catalyse its decomposition It is stored in paraffin wax coated with glass, plastic or teflon bottles. Small amounts of acid, glycerol, alcohol, acetanilide and H3P04 often used as stabilizers to check its decomposition.
Uses
It acts as an antiseptic and a germcide and hence is used for cleaning cuts and wounds. It is used for bleaching delicate articles like wool, hair, feather, ivory etc. It is used as an aerating agents in production of spong rubber. It is used as an antichlor. It finds use in deodorants. It is also used for cleaning ears and removing the surgical dressing. It is an effective antidote for phosphorous and cyanide poisoning.
Potassium Permanganate
Chemical Formula: KMnO2
Molecular Weight: 158.03
It is an inorganic chemical compound having not less than 99.0% of KMnO2. Formerly known as Permanganate of Potash or Candy’s Crystals.
Method of Preparation
On large scale, it is prepared by mixing a solution of KOH with powdered manganese oxide and potassium chlorate in the presence of air or an oxidizing agent.
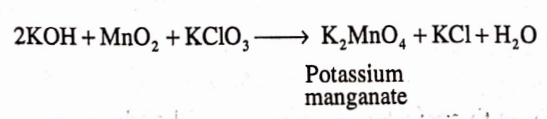
Potassium manganate so formed is extracted along with boiling water and a current of chlorine, C02 or ozonised air is passed into the liquid untill it gets converted to permanganate. The MnO2 formed is removed.

When carbon dioxide is passed through the chlorine solution, manganate gets converted into KMnO4

The solution of KMn04 is drawn off from any precipitate of Mn02 which is then concentrated and crystallized. The crystals are then centrifuged and dried.
Properties
It occurs as odourless dark purple or almost black prismatic crystals or granular powder. It has a sweet and astringent taste. It decomposes with a risk of explosion, in contact with certain organic substances.
It has a specific gravity of about 2.703. It is stable in air. It is soluble in water. When it is heated, it decomposes at a high temperature of about 240°C
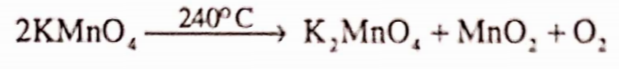
Alkaline or neutral solution of potassium permanganate is able to oxidize iodide to iodate
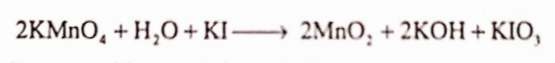
In acidic medium iodine gets liberated from iodates.
Uses
It finds use in the treatment of urethritis. It possesses oxidizing properties and oxidizes proteins and other bioorganic substance. It has the capability to destroy the poison and prevents absorption.
Its solutions are used to clean the ulcer or abscesses, as wet dressings and in baths in eczematous condition. It finds use as an antidote in the case of poisoning by barbiturates, chloral hydrate and many alkaloids.
Chlorinated Lime Or Bleaching Powder Ca(OcI)CI
Chemical Formula : Ca(C1O)2
Molecular Weight: 136.98
Synonyms: Bleaching powder, chloride of lime.
Method of Preparation
It is prepared by the chlorination of slaked lime.
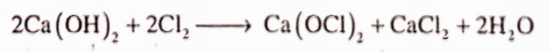
It is usually a mixture of calcium hypochlorite (Ca(OCl)2) and calcium chloride with some slaked lime
Properties
It is dull white powder with characteristic odour. It is slightly soluble in water and in alcohoi. On exposure to air, it becomes moist and gradually decomposes with the loss of chlorine.
Its aqueous solution is strongly alkaline. It is able to oxidize many salts such as manganous salts to permanganate. Carbon dioxide gets absorbed and chlorine is evolved.
Uses
It is also used as an ingredient in bleaching powder, used for bleaching cotton and linen. It is also used in sugar industry for bleaching sugar-cane juice before its crystallization.
It is used as a sanitizer in outdoor swimming pools in combination with a cyanuric acid stabilizer which reduces the loss of chlorine due to ultraviolet radiation. It has the bactericidal action.
Iodine
Chemical Formula: I2
Molecular Weight: 253.8
Iodine I is a dark violet non-metallic halogen element.
Method of Preparation
Iodine is manufactured by extracting kelp (sea weed’s ash) with water. The solution is concentrated when the sulphate and chloride of sodium and potassium get crystallized out, leaving freely soluble sodium and potassium iodides in the mother liquor.
Sulphuric add is added to the mother-liquor which then gets decanted off.
![]()
In the laboratory, it reacts by heating potassium iodide or sodium iodide with dilute sulphuric add and manganese dioxide.
![]()
Properties
It occurs as heavy, bluish-black or greyish violet brittle plates with a metallic lusture. It is very slightly soluble in alcohol, chloroform and slightly soluble in concentrated solutions of iodides, carbon disulphide, solvent ether, carbon tetrachloride, chloroform, give violet solutions.
Uses
It is used as an anti-hyperthyroid. It is used in the manufacture of dyestuffs and drugs. It is used as a reagent in analytical chemistry. It is used in the manufacture of compounds used in Photography.
Prolonged use of iodine may produce metallic taste, increased salivation, burning pain. It can give rise to allergic reactions also.
Silver Nitrate
Chemical Formula: AgNO3
Molecular Weight: 169.89
It contains not less than 99.5% and not more than the equivalent of 100.5% of AgNO3.
Preparation
It is prepared by the reaction of silver with nitric acid which results into the formation of silver nitrate.
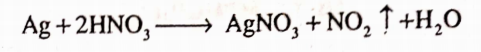
Properties
It occurs as colourless or white, odourless rhombic crystals. It is having a bitter caustic or metallic taste. It is highly soluble in boiling water when exposed to light or organic/blunder matter, it turns into grey or greyish black colour due to its reduction in metallic silver.
Uses
Silver salts have antiseptic properties. It is widely used as an astringent and germicide. Dilute solution of AgNO3 used to be dropped into new bom babies at birth to prevent contraction of gonorrhea from the mother.
Eye infection and blindness of new bom reduced by this method, incorrect damage, however could cause blindness in extreme cases.
Povidone-Iodine (Pv-I)
It is a stable chemical complex of Polyvinyl Pyrfolidonfe (povidone, PVP) and Elemental Iodine. It contains 9.0% to 12.0% available iodine, calculated on a dry basis. It belongs to iodophors class of compounds.
Properties
It is having a slight characteristic odour. It is a yellowish-brown amorphous powder. It is soluble in water and in alcohol but practically insoluble in organic solvents. It is acidic litmus.
Uses
It has a broad spectrum antiseptic for topical application in the treatment and prevention of infection in wounds. It is used as a disinfectant. It is also used as gargles and mouthwashes for the treatment of infections in the oral cavity.
Silver Protein
Chemical Formula: Variable
Molecular Weight : 169.89
Method of Preparation
It is obtained by treating silver salts with an excess of denaturated protein. The product is then dried in vaccum and finally stored in amber coloured bottles.
Properties
It occurs as dark brown or almost black shinning scales or granules. It is odourless and hygroscopic in nature. It is freely soluble in water but almost insoluble in alcohol, chloroform and ether.
When dissolved in water it forms colloidal solutions. It is affected by light and, therefore, should be protected from light. Mild silver protein solution in nitric acid yields a precipitate of silver chloride.
Uses
It is found to be useful in the treatment of infections of the respiratory tract through the prophylactic use which has been discontinued due to harmful nature. It possess to have mild antibacterial properties.
Mercury
Chemical Formula: Hg
Molecular Weight: 200.6
Synonym: Hydragyrum, Quick-silver
The Principle ore of Mercury is a red sulphide mineral called Cinnabar, HgS. It also occur as an amalgam of silver and gold and in small globules through rocks.
Preparation
It is obtained by roasting cinnabar in a current of air.
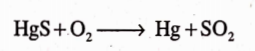
The free mercury gets liberated and further it gets purified by volatilisation or chemically by dropping mercury into a column of dilute nitric acid for removing basic impurities.
Properties
It occurs as shiny, silvery heavy liquid. It gets easily converted into the shape of globules. It readily volatiles on heating, its melting point is -38.9°C.
It is almost insoluble in water, alcohol and hydrochloric acid. It gets easily reacted by centrated sulphuric acid which results into the evolution of sulphuric dioxide and formation of mercuric sulphate.
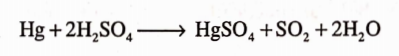
Uses
It is used as a Pharmaceutical Aid. It has Carthatic and Parasitic action. Its solution of salts have violent corrosive effects on skin and mucous membrane, nausea, vomiting, abdominal pain, bloody diarrhoea, kidney damage and death.
Compounds of mercury and its salts have declined greatly during this century.
Note
Avoid frequent or prolong contact of mercury with the skin. Mercury spills (zuhether in laboratory or in house from broken thermometer etc.) should be handled and collected with special care.
Yellow Mercuric Oxide
Chemical Formula: HgO
Molecular Weight: 216.16
Preparation
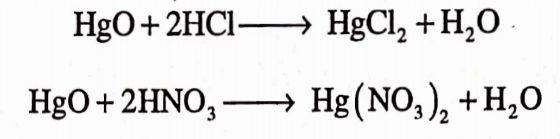
It is prepared by precipitation of mercuric nitrate or mercuric chloride with sodium hydroxide solution. The yellow precipitate obtained gets settle down and further washed by decantation.
It gets filtered and re-washed to separate chloride and further gets dried off in air.

As the yellow precipitates gets affected by light, that is if all the reactions should be carried out in dark to maintain its orange yellow colour.
Properties
It is odourless orange yellow amorphous powder. Its colour varies from orange to yellow according to the temperature at which it is prepared. It gets decolourised on exposure to light. It is insoluble in water and alcohol but soluble in acids sch as HCl or H2SO4.
Uses
It has been used as a topical antiseptic, in conjunctivitis and in chronic ulcers. Due to its insolubility, it slowly releases Hg2+ ions which has mild sustained local antibacterial properties.
It is used in eye ointments for the local treatment of minor infections and inflammation of the eye including conjunctivitis.
Ammoniated Mercury
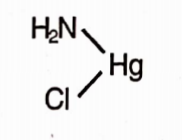
Chemical Formula: NH2HgCl
Molecular Weight: 252.1
Synonym
Aminochloride of mercury, White precipitates.
Preparation
It is prepared by adding an aqueous solution of mercury chloride to ammonia solution with constant stirring. The precipitates so obtained gets filtered off and then washed with cold water to remove ammonium chloride. It is dried at a temperature not exceeding 30°C.
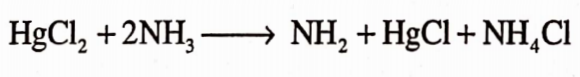
Properties
It is a white heavy amorphous powder. It is odourless. It is almost insoluble in water, alcohol and ether but it is readly soluble in warm acetic acid. It is stable in air. It darkens on exposure to light.
It gets decomposed when dissolved in cold water. In boiling water, it gets hydrolyzed to yellow basic compound :- NH2HgCl; HgO
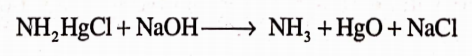
Uses
It is used in the treatment of skin diseases such as impetigo, in crab infections. Adverse effects include allergic reactions and mercury poisoning
Zinc Undecylenate
Chemical Formula : [CH2 = CH (CH2 )8 COO]2 Zn
Molecular wt: 431.9
Synonyms
Zinc Undecenoate, Zinc undec-10-enoate, Zinc undecyl.
Preparation
It is prepared by precipitation from a solution of sodium salt.
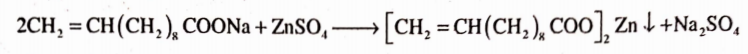
Properties
It is a fine white powder. It is stable and is practically insoluble in water and in alcohol.
Uses
It is topical antifungal agent.
Sodium Antimony Gluconate
Chemical Formula: C6H8NaO7Sb
Molecular Weight:‘336.88
Synonym: Antimony Sodium Gluconate
Method of Preparation
It is prepared by the action of sodium hydroxide on antimony trichloride and a solution of gluconic acid.
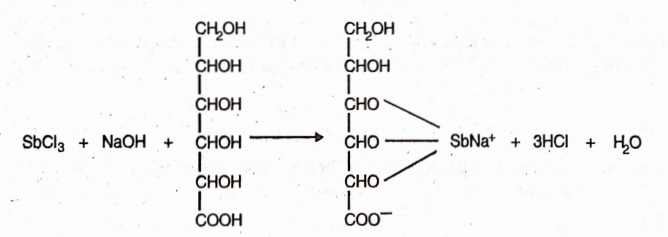
Properties
It is an odourless, white amorphous powder. It is soluble in water but insoluble in alcohol and solvent ether. Solution of Antimony Gluconate should be prepared immediately before use.
Uses
It is used in the treatment of leishmaniasis (Kala-azar) and as antischistosomal.
Antimicrobials Short Answer Questions
Question.1. Define Antimicrobials?
Answer. An antimicrobial (disinfectant and antiseptic) is a substance that kills or inhibits the growth of microorganism such as bacteria, fungi or protozoans.
Question.2. Classify antimicrobials on the basis of the exact mode or mechanism of action?
Answer.
- Antiseptic Bacteriostatics
- Disinfectant Sanitizers
- Germicides
Question.3. Define Antiseptic?
Answer. Antiseptic is a substance that prevents or arrests the growth of action of microorganisms.
Question.4. How do antiseptics act?
Answer. Antiseptics act by inhibiting the activity of microorganism or destroying them especially of agents applied to living tissues like surface of skin, application of antiseptic dressing on wounds.
Question.5. Write down the two examples of antiseptics?
Answer. Iodine, Boric acid, Cetrimide.
Question.6. Define Disinfectants?
Answer. Disinfectants are the drugs or substances used either to kill bacteria or prevent their growth or multiplication.
Question.7. Write down the two examples of disinfectants?
Answer. Cresol, Phenol.
Question.8. What is the difference between antiseptics and disinfectants?
Answer.
Antiseptics
- It is a substance that prevents or arrests the growth of microorganism.
- It is applied on living tissues.
Disinfectants
- It is a substance used either to kill bacteria or prevent their growth or multiplication. ,
- It is applied on non-living surfaces.
Question.9. What do you understand by the term Germicide? How do they act?
Answer. Germicides are the substances which kill micro-organisms.
Germicide act by oxidation of bacteria protoplasm, by denaturation of bacteria, enzymes and proteins.
Question.10. What is Bacteriostatic?
Answer. These are the substances which primarily function by inhibiting the growth of bacteria. Thus, bacteriostatic agents or drug do not kill but arrest the growth of bacteria.
Question.11. What do you understand by ‘Sanitizers’?
Answer. Sanitizer is the process of rendering sanitary by reducing the number of bacterial contaminants.
Question.12. Enlist various inorganic compounds that are used as Anitmicrobials?
Answer. Boric add, Borax, Hydrogen peroxide, Potassium permanganate, Chlorinated lime,Iodine.
Question.13. What is the chemical formula of Boric acid?
Answer. Chemical formula of Boric add is H3BO3.
Question.14. What is the method of preparation of Boric Acid?
Answer. Boric add is prepared by reacting hydrochloric or sulphuric add with the native borax to product orthoboric add.
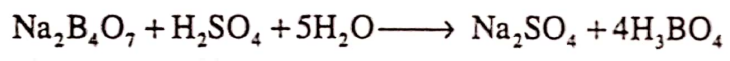
The solution is then filtered off and the crystals obtained are washed and then allow to dry at room temperature.
Question.15. What is the chemical formula of Borax? By which other names they are known in the market?
Answer.
Borax: Na2B4O7.10H2O
Synonyms: Sodium borate, Sodium tetraborate, Sodium pyroborate
Question.16. What is the method of preparation of Hydrogen Peroxide?
Answer: Hydrogen peroxide is prepared by Marck’s process. It is prepared by adding calculated amounts of sodium peroxide to ice cold dilute (20%) solution of H2SO4.
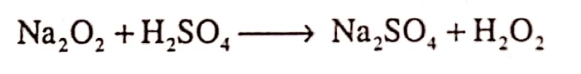
Question.17. Enlist the pharmaceutical uses of Hydrogen Peroxide?
Answer. Hydrogen peroxide is used as an antiseptic and a germicide. It is also used for cleaning cuts and wounds. It is an effective antidote for phosphorous and cyanide poisoning.
Question.18. What is the chemical formula and method of preparation for Potassium permanganate?
Answer. Chemical formula of potassium permanganate is KMnO4.
Potassium permanganate is prepared by mixing a solution of KOH with powdered manganese oxide and potassium chlorate in the presence of air or an oxidising agent and the mixture gets boiled.
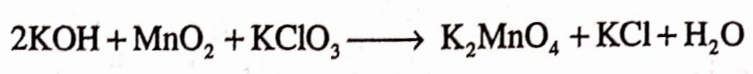
Question.19. What are the Pharmaceutical uses of Potassium Permaganate?
Answer. Potassium permaganate solution is used to clean the ulcer or abcesses, as wet dressings and in baths of eczematous condition
Question.20. What is the chemical formula of Chlorinated Lime?
Answer. Ca(ClO)2
Question.21. What are the uses of Iodine?
Answer. Iodine is used as an antihyperthyroid. It is also used in the manufacture of dye stuffs and drugs.
Question.22. What is the method of preparation of Iodine in laboratory?
Answer. In laboratory, Iodine reacts by heating potassium iodide or sodium iodide wth dilute sulphuric acid and manganese dioxide.
Question.23. What is the general formula of Silver nitrate?
Answer. AgNO3
Question.24. What is the method of preparation of Silver nitrate?
Answer. Silver nitrate is prepared by the reaction of silver with nitric acid which result in the formation of silver nitrate.
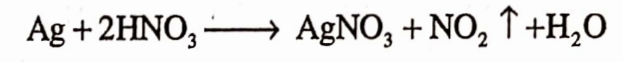
Question.26. What are the other names of Mercury?
Answer. Hyedragyrum, Quick-silver.
Question.27. How do we prepare Mercury?
Answer. Mercury is obtained by roasting cinnabar in a current of air.
The free mercury gets librated and further it gets purified by volatilisation.
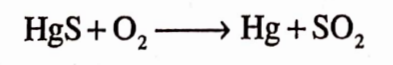
Question.28. What is the chemical formula of yellow mercuric oxide?
Answer. HgO.
Question.29. What is the chemical formula of Ammoniated Mercury?
Answer. NH2HgCl.
Question.30. How do we prepare ammoniated mercury?
Answer. Ammoniated mercury is prepared by adding an aqueous solution of mercury chloride to ammonia solution with constant stirring. The precipitates are obtained.
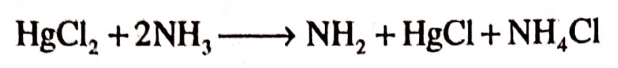
Question.31. How do antimicrobials act?
Answer. Antimicrobial act by either oxidation, halogenation and protein precipitation.
Antimicrobials Fill In The Blanks
1. Antimicrobial is a substance that……………………
Answer: Kills or inhibits bacteria
2. Antimicrobial agents act by ………………….
Answer: Oxidation, halogenation and protein precipitation
3. Antiseptic are applied to…………… while disinfectants are applied to……………….
Answer: Living tissues, non-living objects
4………………..is the process of rendering sanitary by reducing the number of bacterial contaminants.
Answer: Sanitization
5. KMnO4can be used as an…………………………
Answer: Bactericidal, fungicidal, in the treatment of urethritis
6. KMnO4 is………………… reducing agents.
Answer: Strong oxidizing agent
7. Povidone-iodine is a member of class compound referred to as…………………………….
Answer: Iodophors
8. Povidone-Iodine is……………………………
Answer: Polyvinyl pyrollidine
9. Solution of AgNO3 can be used as an………………………
Answer: To prevent contraction of gonorrhea
10. Chemical formula of Ammoniated mercury is……………………
Answer: NH2HgCl
11. Ammoniated mercury is also known as…………………………
Answer: Aminochloride of mercury
12. …………………….include antiseptic, germdde and- disinfectant agent as well as sanitization process.
Answer: Iodine
13………………… give rise to hyperthyroidism symptoms of which are extreme fatigue,goitre.
Answer: Iodine
14. Diluted hydrogen peroxide (3% and 12%) is used to bleach human hair when mixed with ammonia and is called as…………………….
Answer: Peroxide blonde
15. Potassium permaganate is manufactured on a large scale by heating…………………. in the presence of air or as an oxidizing agent such as
Answer: Potassium hydroxide with manganese oxide, potassium nitrate
16. Yellow mercuric oxide is prepared by precipitation of………………………………..with………………….
Answer: Mercuric nitrate with sodium hydroxide
17. Mercury compounds as antimicrobial agents are . …………………..
Answer: Ammoniated mercury, yellow mercuric oxide
18. The chemical formula of Ammoniated Mercury is……………………..
Answer: NH2HgCl
19. Ammoniated mercury is also known as ………………………….
Answer: Aminochloride of mercury, white precipitates
20 ………………………..is prepared by precipitation of a solution of sodium salt.
Answer: Zinc undecylenate
21. The chemical formula of Sodium Antimony Gluconate is……………………..
Answer: C6H8NaO7Sb
22. Antimony gluconate is used as………………..
Answer: Antischistosomal
23……………………is prepared by reacting hydrochloric or sulphuric add. with the native borax to produce orthoboric add.
Answer: Boric acid
24. Hydrogen peroxide is…………………….solution.
Answer: Aqueous
25. ………….is an effective antidote for cyanide poisoning which is also used as an antimicrobial.
Answer: Hydrogen Peroxide
