Alcohol Phenol And Ether
Question 1. Classify the following as primary, secondary, and tertiary alcohols:
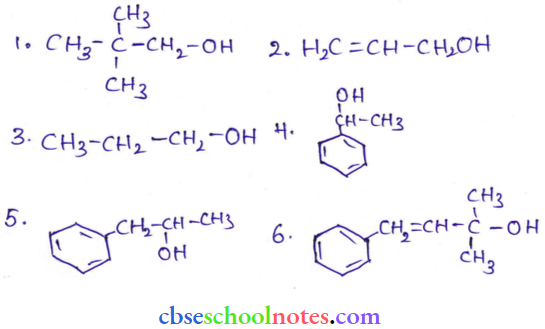
Answer:
- Primary alcohol > (1), (2), (3)
- Secondary alcohol > (4), (5)
- Tertiary alcohol > (6)
Question 2. Identify allylic alcohols in the above examples.
Answer:
- Allylic that means C = C – C – OH
- The alcohols given in (2) and (6) are allylic.
Question 3. Name the following compounds according to the IUPAC system.
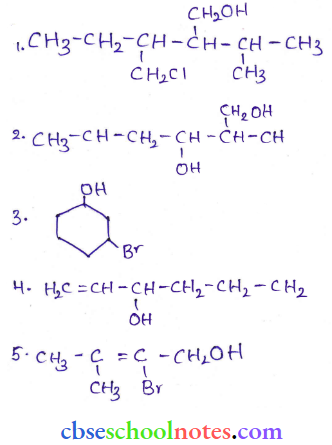
Answer:
- 3-Chloromethyl-2-isopropylpentan-1-ol
- 2, 5 – Dimelhylhcxane – 1,3- cliol
- 3-Bromocyclohexanol
- Hex-1 -en-3-ol
- 2-Bromo-3-methylbut-2-en-1 -ol
Question 4. Show how are the following alcohols prepared by the reaction of a suitable Grignard reagent on methanal.
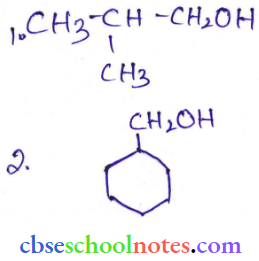
Answer:
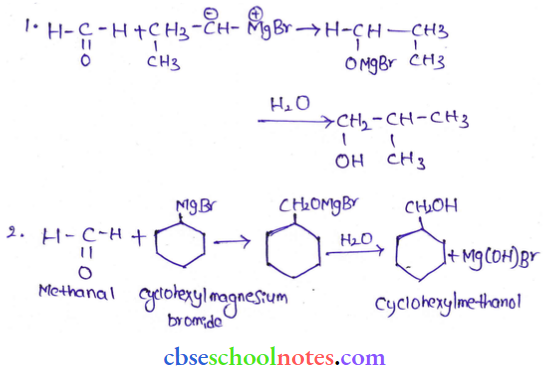
Read and Learn More Class 12 Chemistry with Answers Chapter Wise
Question 5. Write structures of the products of the following reactions:
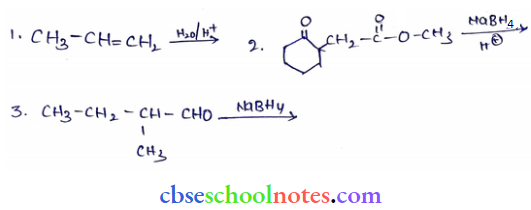
Answer:
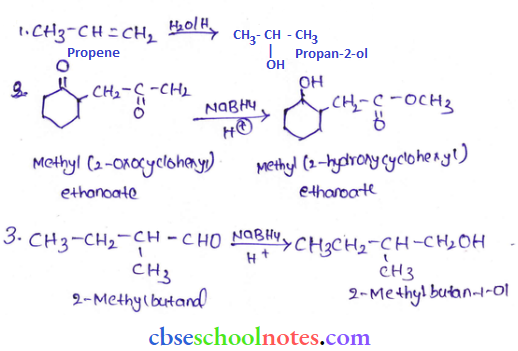
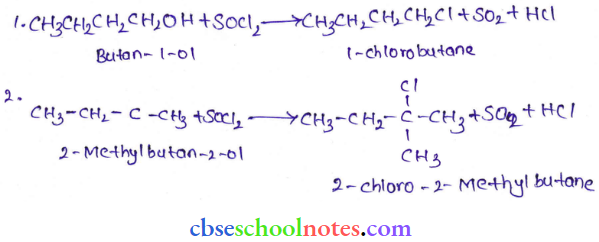
Question 6. Give structures of the products you would expect when each of the following alcohols reacts with
- HCl-ZnCl2
- HBr and
- SOCl2
- Bulan-1-ol
- 2-Methylbutan-2-ol
Answer:
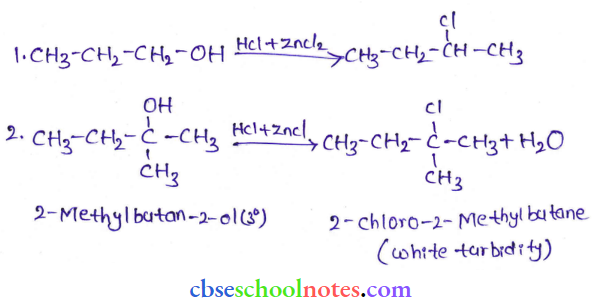
Tertiary alcohols react immediately with Lucas’ reagent.
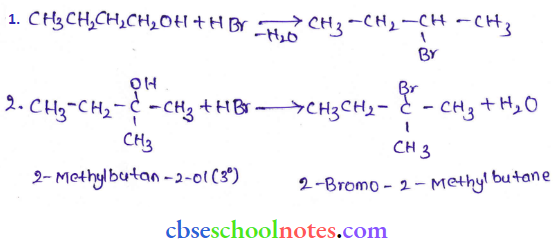
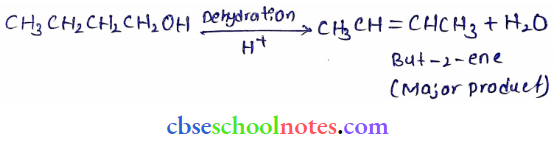
Question 7. Predict the major product of acid-catalyzed dehydration of
- 1-methylcyclohexane and
- Butan-1-ol
Answer:
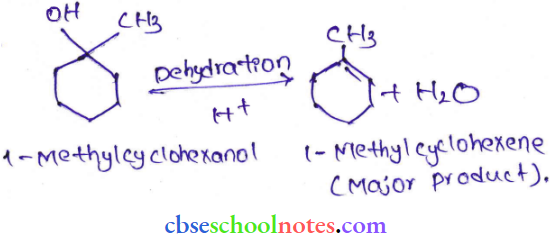
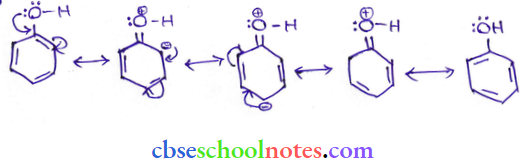
Question 8. Ortho para nitrophenols are more than phenol. DFrawn the resonance structure of the corresponding phenoxide ions.
Answer:
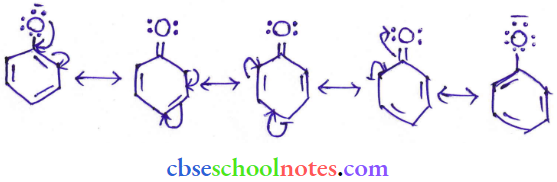
(Resonance structure of the phenoxide ion).
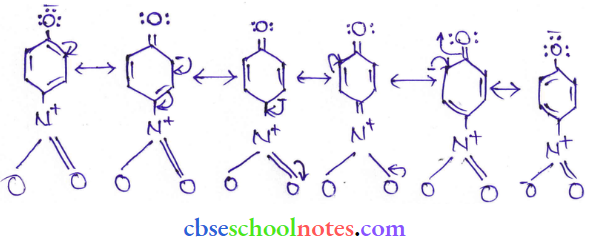
(Resonance structure of p-nitro phenoxide ion)
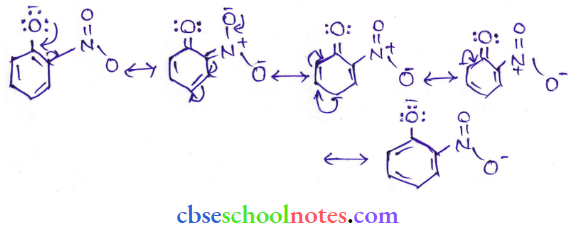
(Resonance structure of o-nitro phenoxide ion)
It can be observed that the presence of the nitro group increases the stability of phenoxide ions.
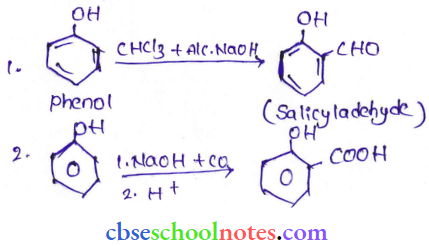
Question 9. Write the equations involved in the following reactions:
- Reimer – Tiemann reaction
- Kolbe – Schmidt reaction
Answer:
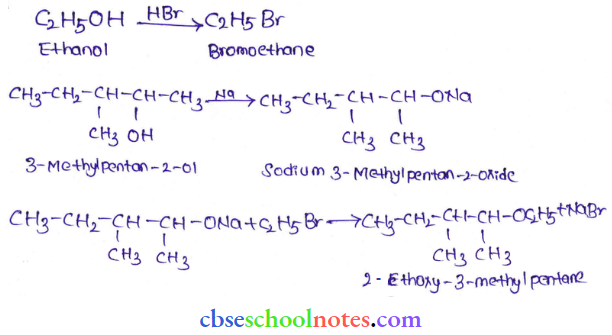
Question 10. Write the reactions of Williamson synthesis of 2-ethoxy-3-inethylpentane starting from ethanol and 3-methylpentan-2-ol.
Answer:
Question 11. Which of the following is an appropriate set of reactants for the preparation of 1-methoxy-4- nitrobenzene and why?
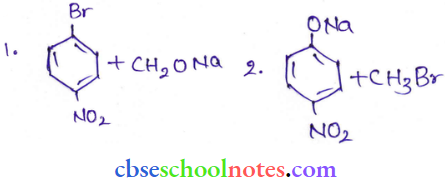
Answer: Set (2) is an appropriate set of reactants for the preparation of 1-methoxy-4-nitrobenzcne.

In set (1), the Bromine atom is stable by resonance.
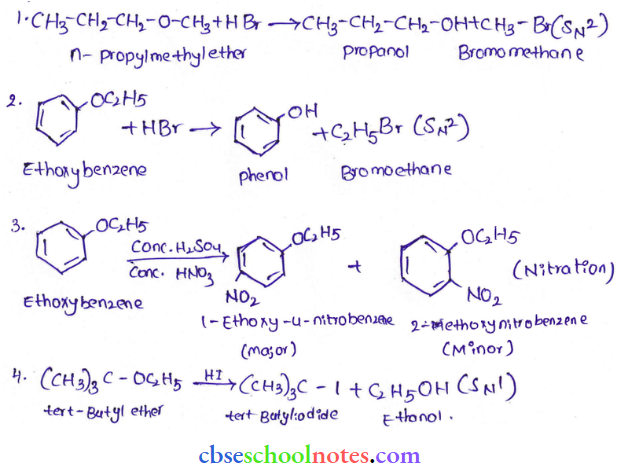
Question 12. Predict the products of the following reactions:
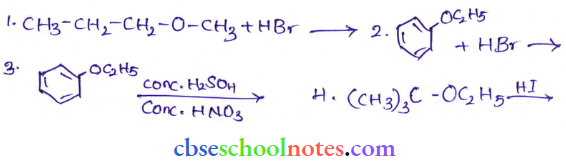
Answer:
Question 13. Give IUPAC names of the following compounds:
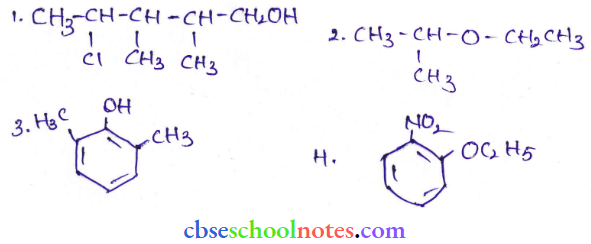
Answer:
- 4-ChIoro-2,3-dimethylpentan-1-oI
- 2-Ethoxy propane
- 2,6-Diinethylphenol
- 1-Elhoxy-2-nilrocyclohexane
Question 14. You will notice that the reaction produces a primary alcohol with methanol, a secondary alcohol with other aldehydes, and a tertiary alcohol with ketones. Give the structures and IUPAC names of the products expected from the following reactions:
- Catalytic reduction of butane.
- Hydration of propene in the presence of dilute sulphuric acid.
- Reaction of propanone with methyl magnesium bromide followed by hydrolysis.
Answer:
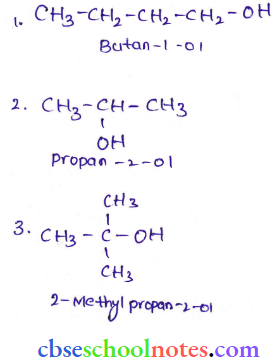
Question 15. Arrange the following sets of compounds in order of their increasing boiling points:
- Pentan-1-ol, butan-1-ol, butan-2-ol, ethanol, propan-1-ol, methanol.
- Pentan-1-ol, n-butane, pentanal, ethoxyethane.
Answer:
- Methanol, ethanol, propan-1-ol, butan-2-ol, butan-1-ol, pentan-1-ol.
- n-Butane, ethoxyethane, pentanal, and pentan-1-ol.
Question 16. Arrange the following compounds in increasing order of their acid strength: Propan-1-ol, 2, 4, 6-trinitrophenol, 3-nitrophenol, 3,5-dinitrophenol, phenol, 4-methylphenol.
Answer: Propan-1-ol, 4-methylphenol, phenol, 3-nitrophenol, 3, 5-dinitrophenol, 2, 4, 6-trinitrophenol.
Question 17. Write the structures of the major products expected from the following reactions:
- Mononitration of 3-methyl phenol
- Denitration of 3-methyl phenol
- Mononitration of phenyl methanoate.
Answer: The combined influence of -OH and -CH3 groups determines the position of the incoming group.
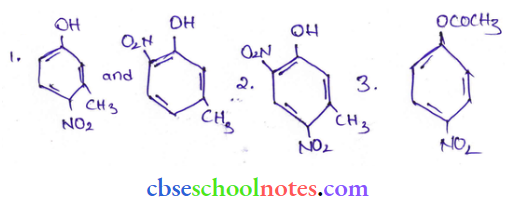
Question 18. The following is not an appropriate reaction for the preparation of t-butyl ethyl ether.
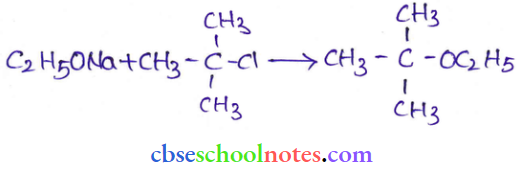
- What would be the major product of this reaction?
- Write a suitable reaction for the preparation of t-butyl ethyl ether.
Answer:
1. The major product of the given reaction is 2-methyl prop-1-one.
This is because sodium ethoxide is a strong nucleophile as well as a strong base. Thus elimination reaction predominates over substitution.
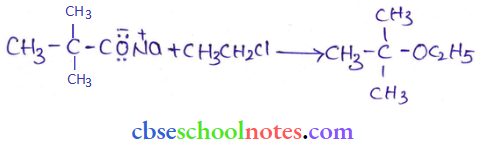
2. Phenols are also converted to ethers by this method. In this, phenol is used as the phenoxide moiety.
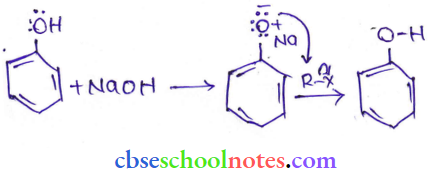
Question 19.
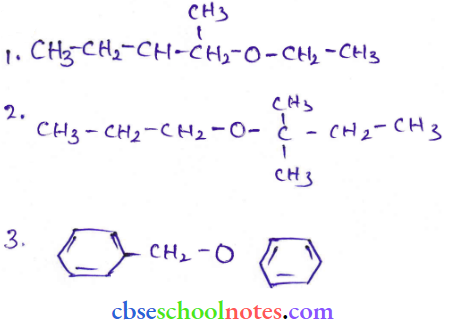
Answer:
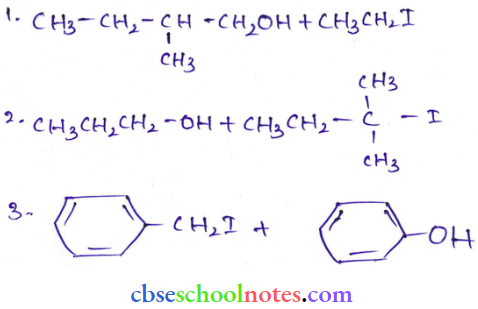
Question 20. Write IUPAC names of the following compounds:
Answer:
- 2, 2, 4-Trimethylpcntan-3-ol
- 5-Ethylheplane-2, 4-diol
- Butane-2, 3-diol
- Propane-1, 2, 3-triol
- 2-Methylphenol
- 4-Methylphenol
- 2. 5-Dimcthylphenol
- 2, 6-Dimethylphenol
- 1-Methoxy-2-methylpropane
- Ethoxyben/ene
- 1-Phenoxyheptane
- 2-Elhoxvbutane
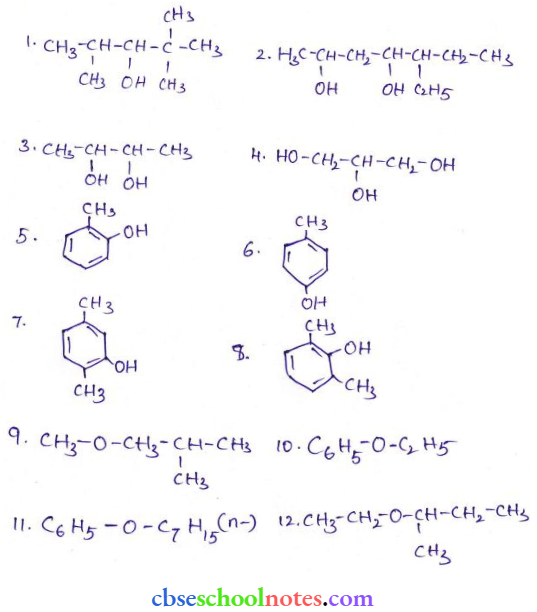
Question 21. Write structures of the compounds whose IUPAC names are as follows:
Answer:
- 2-Methylbutan-2-ol
- 1-Phenyl-2-ol
- 3,5- Dimethylhexene-1,3,5-triol
- 2,3-Diethylphenol
- 1-Ethoxypropane
- 2-Ethoxy-3-methyl pentane
- Cyclohexylmethanol
- 3-Cyclohexylpentan-3-ol
- Cyclopent-3-en-1-ol
- 3-Chloromethylpentan-1-ol
Answer:
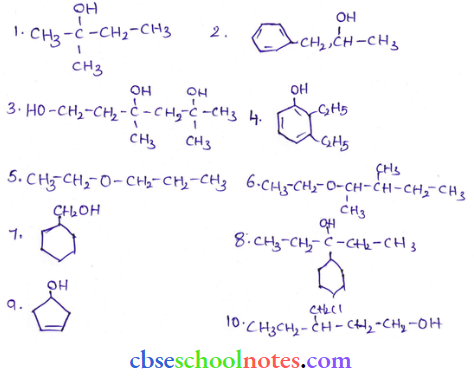
Question 22. Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names. Classify them as primary, secondary, and tertiary alcohols.
Answer:
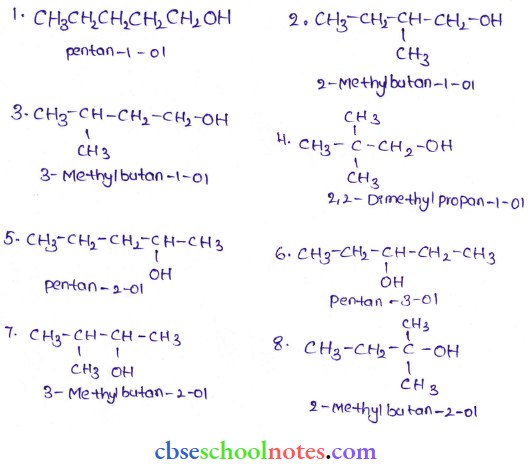
- Primary alcohols – (1), (2)(3), and (4)
- Secondary alcohols – (5),(6) and (7)
- Tertiary alcohol – (8)
Question 23. Explain why propanol has a higher boiling point than that of the hydrocarbon, butane.
Answer:
Propanol undergoes intermolecular H-bonding because of the presence of the -OH group. On the other hand, butane does not
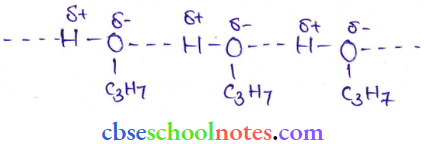
Question 24. Explain why are alcohols comparatively more soluble in water than the corresponding hydrocarbons.
Answer:
Hydrogen bonding exists between alcohol and water molecules while hydrocarbons are nonpolar, they can not form an H-bond with water molecules.
Question 25. What is meant by hydroboration oxidation reaction? Illustrate it with an example.
Answer:
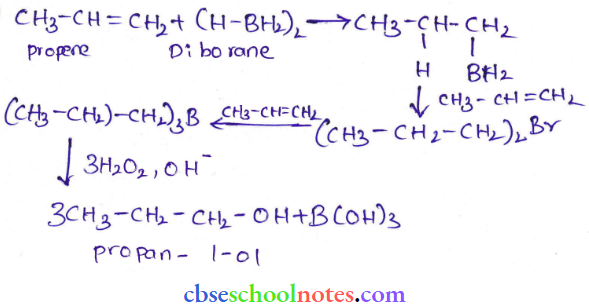
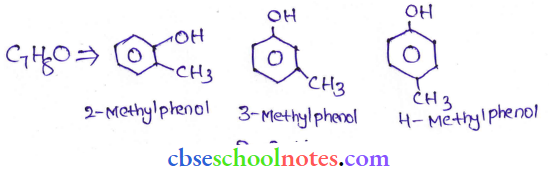
Question 26. Give the structures and IUPAC names of monohydric phenols of molecular formula C7H8O.
Answer:
Question 27. While separating a mixture of ortho and para nitrophenols by steam distillation, name the isomer which is steam volatile. Give reason.
Answer: O-nitrophenol is steam volatile due to intramolecular hydrogen bonding.
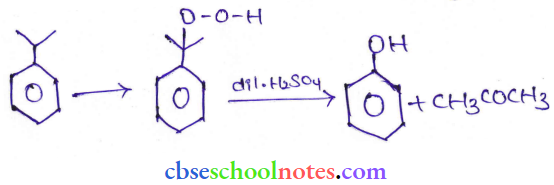
Question 28. Give the equation of reaction of preparation of phenol from cumene.
Answer:
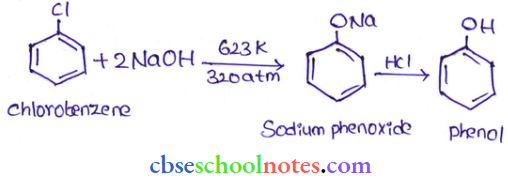
Question 29. Write a chemical reaction for the preparation of phenol from chlorobenzene.
Answer:
Chlorobenzene is fused with NaOH (at 623 K and 320 atm pressure) to produce sodium phenoxide, which gives phenol on acidification.
Question 30. You are given a benzene, cone. H2SO4 and NaOH. Write the equations for the preparation of phenol using these reagents.
Answer:

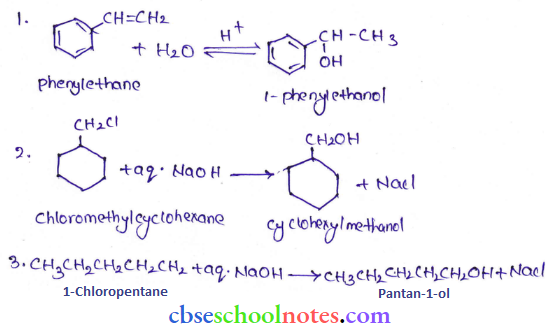
Question 31. Show how will you synthesize:
- 1-phenylethanol from a suitable alkane.
- CyelohexyI methanol using an alkyl halide by an SN2 reaction
- Pentan-1-ol using a suitable alkyl halide?
Answer:
Question 32. Give two reactions that show the acidic nature of phenol. Compare the acidity of phenol with that of ethanol.
Answer:
The acidic nature of phenol can be represented by the following two reactions.
1. Phenol reacts with sodium to give sodium phenoxide, liberating H2
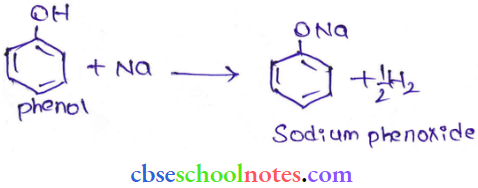
2. Phenol reacts with sodium hydroxide to give sodium phenoxide and water as by-products.

The acidity of phenol is more than that of ethanol. This is because after losing a proton, the phenoxide ion undergoes resonance and gets stabilized whereas the ethoxide ion does not.
Question 33. Explain why is ortho-nitro phenol more acidic than ortho-methoxy phenol.
Answer: Electron withdrawing effect of nitro group and electron releasing effect of methoxy group.
Question 34. Explain how the -OH group attached to a carbon of benzene ring activates it towards electrophilic substitution.
Answer: Electron density at the o- and p-position increases and thus the electrophile attacks easily.
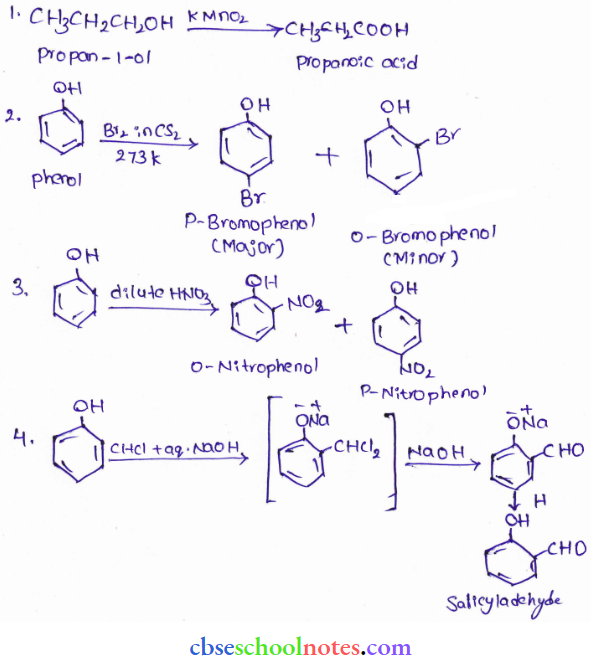
Question 35. Give equations of the following reactions:
- Oxidation of propane-1 -ol with KMnO4 solution.
- Bromine is CS2 with phenol.
- Dilute HNO3 with phenol.
- Treating phenol with chloroform in the presence of aqueous NaOH.
Answer:
Question 36. Explain the following with an example.
- Williamson ether synthesis.
- Unsymmetrical ether.
Answer:
1. Williamson’s ether synthesis:
Williamson ether synthesis is a laboratory method to prepare symmetrical and unsymmetrical ethers by allowing alkyl halides to react with sodium alkoxide.

This reaction involves an SN2 attack of the alkoxide ion on the alkyl halide. Better results are obtained in the case of primary alkyl halides.
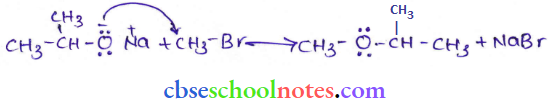
If the alkyl halide is secondary or tertiary, then elimination competes over substitution.
2. Unsymmetrieal ether:
An unsymmetrical ether is an ether where two alkyl groups on the two sides of an oxygen atom differ. For example: ethyl methyl ether (CH3 -O-CH2CH3).
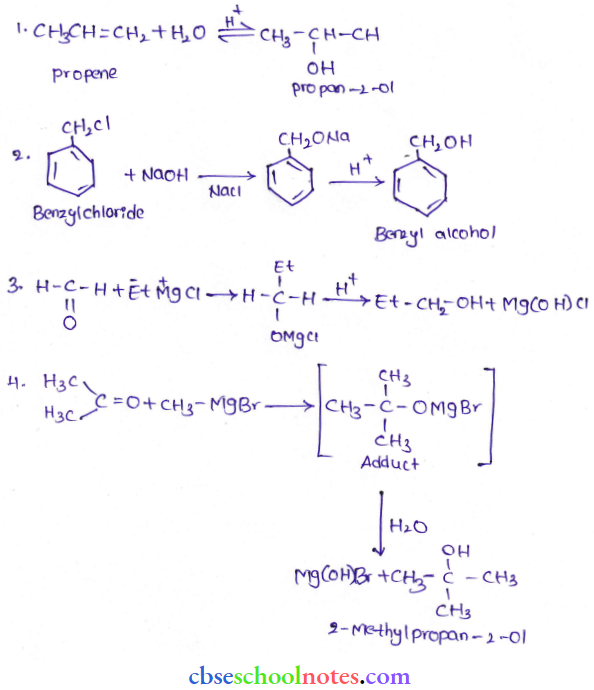
Question 37. How are the following conversions carried out?
- Propene → Propan-2-ol
- Benzyl chloride → Benzyl alcohol
- Ethyl magnesium chloride → Propan-1-ol.
- Methyl magnesium bromide → 2-Methylpropan-2-ol,
Answer:
Question 38. Give a reason for the higher boiling point of ethanol in comparison to methoxyrnethane.
Answer:
- Ethanol undergoes intermolecular H-bonding due to the presence of the -OH group, resulting in the association of molecules.
- On the other hand, methoxyrnethane does not undergo H-bonding. Hence, the boiling point of ethanol is higher than that of methoxyrnethane.
Question 39. Illustrate with examples the limitations of Williamson synthesis for the preparation of certain types of ethers.
Answer: The reaction of Williamson synthesis involves an SN2 attack of an alkoxide ion on a primary alkyl
halide.
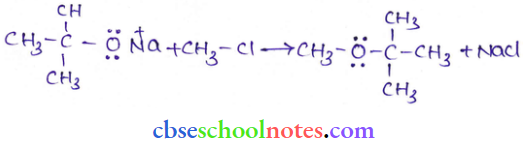
- But if secondary or tertiary alkyl halides are taken in place of primary alkyl halides, then elimination would compete over substitution. As a result, alkenes would be produced.
- This is because alkoxides are nucleophiles as well as strong bases. Hence, they react with alkyl halides, which results in an elimination reaction.

Question 40. How is 1-propoxypropane synthesized from propan- 1-ol? Write the mechanism of this reaction.

Answer:
The mechanism of this reaction involves the following three steps:
Step 1: Protonation
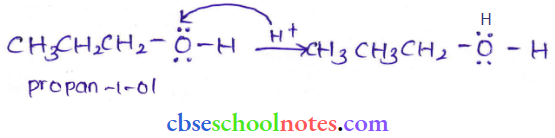
Step 2: Nucleophilic attack
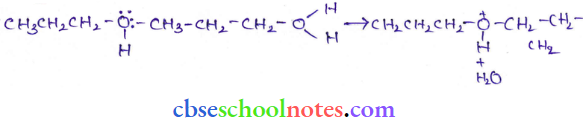
Step 3: Deprotonation
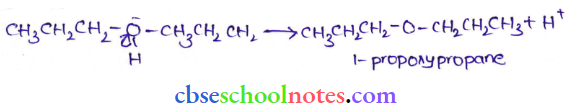
Question 41. The preparation of ethers by acid dehydration of secondary or tertiary alcohols is not a suitable method. Give reason.
Answer:
- The formation of ethers by dehydration of alcohol is a bimolecular reaction (SN2) involving the attack of an alcohol molecule on a protonated alcohol molecule.
- In this method, the alkyl group should be unhindered. In the case of secondary or tertiary alcohols, the alkyl group is hindered. As a result, elimination dominates substitution. Hence, in place of others, alkenes are formed.
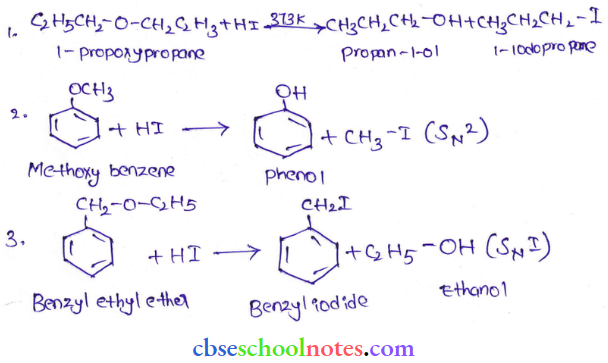
Question 42. Write the equation of the reaction of hydrogen iodide with:
- 1-propoxypropane
- Methoxybcnezcnc and
- Benzyl ethyl ether
Answer:
Question 43. Explain the fact that in aryl alkyl ethers
- The alkoxy group activates the benzene ring towards electrophilic substitution and
- It directs the incoming substituents to ortho and para positions in the benzene ring.
Answer:
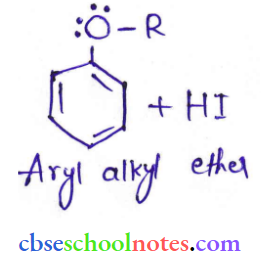
In arly alkyl ethers, due to the + R effect of the alkoxy group, the electron density in the benzene ring increases as shown in the following resonance structure.
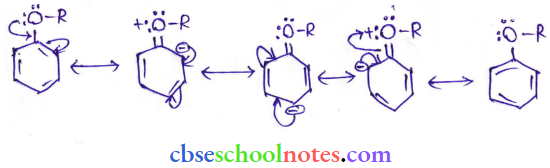
Thus, benzene is activated towards electrophilic substitution by the alkoxy group.
2. It can also be observed from the resonance structures that the electron density increases more at the ortho and para positions than at the meta position. As a result, the incoming substituents are directed to the ortho and para positions in the benzene ring.
Question 44. Write the mechanism of the reaction of III with methoxymethyl.
Answer:
The mechanism of the reaction of III with methoxymethane involves the following steps:
Step 1: Protonation of methoxymelthane:
![]()
Step 2: Nucleophilic attack of I–

Question 45. Write equations of the following reactions:
- Friedel-Crafts reaction-alkylation of anisole.
- Nitration of anisole.
- Bromination of anisole in ethanoic acid medium.
- Friedcl-Craft’s acetylation of anisole.
Answer:

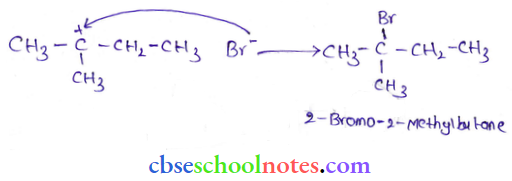
Question 46. When 3-methyl butane-2-ol is treated with HBr, the following reaction takes place:

Give a mechanism for this reaction.
Answer: The mechanism of the given reaction involves the following steps:
Step 1: Protonation
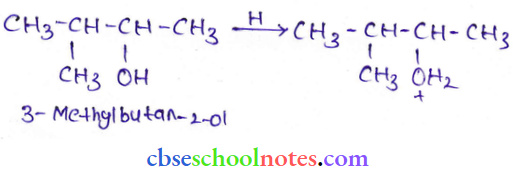
Step 2: Formation of 2° carbonation by the elimination of a water molecule
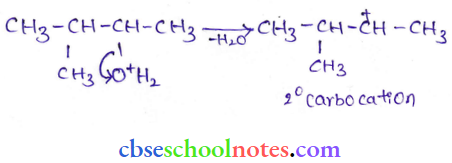
Step 3: Re-arrangement by the hydride-ion shift

Step 4: Nucleophilic attack