Biomolecules
Question 1. Glucose or succrose are soluble in water but cyclohexane or benzene (simple six-membered ring compounds) are insoluble in water. Explain.
Answer:
- A glucose molecule contains five -OH groups while a sucrose molecule contains eight -OH groups. Thus, glucose and sucrose undergo extensive H-bonding with water.
- Hence, they are soluble in water.
- But, cyclohexane and benzene do not contain -OH groups. Hence, they cannot undergo II- bonding with water and as a result, they are insoluble in water.
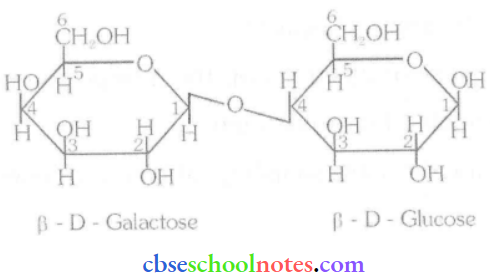
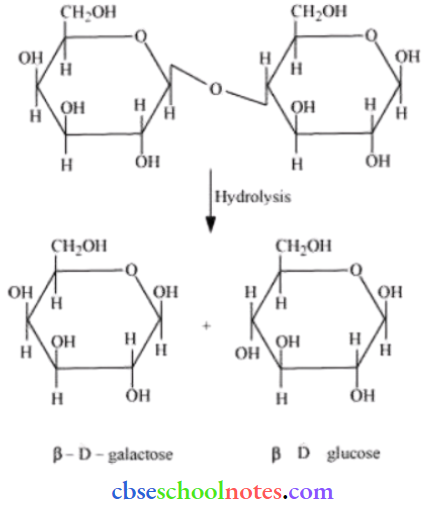
Question 2. What are the expected products of the hydrolysis of lactose?
Answer:
Lactose is composed of β-D galactose and β-D glucose. Tims, on hydrolysis, gives β-D galactose and β-D glucose.
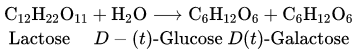
Question 3. How do you explain the absence of an aldehyde group in the pentaacctate of D-glucose?
Answer:
D-glucose reacts with hydroxylamine (NH2, OH) to form an oxime because of the presence of an aldehydic (-CHO) group of carbonyl carbon. This happens as the cyclic structure of glucose forms an open chain structure in an aqueous medium, which then reacts with NH2, OH to give an oxime.
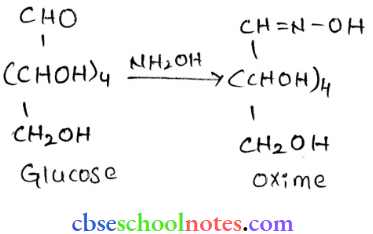
But pentaacctate of D-glucose does not react with NH2, OH. This is because pentaacctate does not form an open chain structure.
Read and Learn More Class 12 Chemistry with Answers Chapter Wise
Question 4. The melting points and solubility in water of amino acids are generally higher than those of the corresponding halo acids. Explain.
Answer:
Both acidic (carboxyl) as well as basic (amino) groups are present in the same molecule of amino acids. In aqueous solutions, the carboxyl group can lose a proton and the amino group can accept, thus giving rise to a dipolar ion known as a zwitter ion.
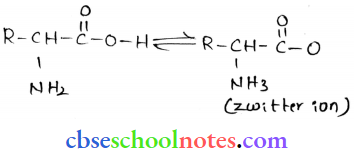
- Due to this dipolar behaviour, they have strong electrostatic interactions within them and with water. However, halo-acids do not exhibit such dipolar behaviour.
- For this reason, the melting points and the solubility of amino acids in water are higher than those of the corresponding halo acids.
Question 5. Where does the water present in the egg go after boiling the egg?
Answer:
When an egg is boiled, the proteins present inside the egg get denatured and coagulate. After boiling the egg. the water present in it is absorbed by the coagulated protein through H-bonding.

Question 6. Why cannot vitamin C be stored in our bodies?
Answer: Vitamin C cannot be stored in our body because it is water-soluble. As a result, it is readily excreted in the urine.
Question 7. What products would be formed when a nucleotide from DMA containing thymine is hydrolysed?
Answer: When a nucleotide from the DNA containing thymine is hydrolyzed, thymine. β-D-2- deoxyribose and phosphoric acid are obtained as products.
Question 8. When RNA is hydrolysed, there is no relationship among the quantities of different bases obtained. What does this fact suggest about the structure of RNA?
Answer:
- A DNA molecule is double-stranded in which the pairing of bases occurs. Adenine always pairs with thymine, while cytosine always pairs with guanine.
- Therefore, on hydrolysis of DNA. the quantity of adenine produced is equal of that of thymine and similarly, the quantity of cytosine is equal to that of guanine.
- But when RNA is hydrolyzed, there is no relationship among the quantities of the different bases obtained. Hence, RNA is single-stranded.
Question 9. What are monosaccharides?
Answer:
- Monosaccharides are carbohydrates that cannot be hydrolysed further to give simpler units of polyhydroxy aldehyde or ketone. Monosaccharides are classified based on a number of carbon atoms and the functional group present in them.
- Monosaccharides containing an aldehyde group are known as aldose and those containing a keto group are known as ketose.
- Monosaccharides are further classified as triose, tetrose, pentose, Itexose, and heptose according to the number of carbon atoms they contain. For example, a ketose containing 3 carbon atoms is called ketotriose and an aldose containing 3 carbon atoms is called aldotriose.
Question 10. What are reducing sugars?
Answer: Reducing sugars are carbohydrates that reduce l’ehling’s solution and Tollen’s reagent. All monosaccharides and disaccharides, excluding sucrose, are reducing sugars.
Question 11. Write two main functions of carbohydrates in plants.
Answer:
Two main functions of carbohydrates in plants are:
- Polysaccharides such as starch serve as storage molecules.
- Cellulose, a polysaccharide, is used to build the cell wall.
Question 12. Classify the following into monosaccharides and disaccharides. Ribose, 2-deoxyribose. maltose, galactose, fructose and lactose
Answer:
- Monosaccharides: Ribose, 2-deoxyribose, galactose, fructose
- Disaccharides: Maltose, lactose
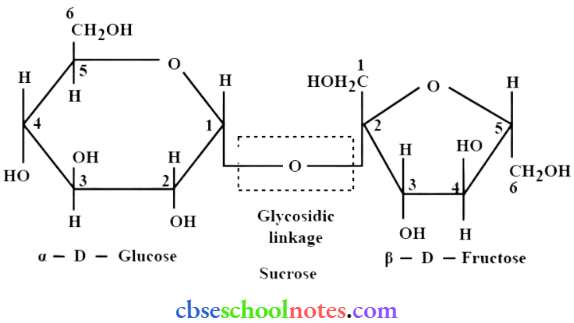
Question 13. What do you understand by the term glycosidic linkage?
Answer:
- Glycosidic linkage refers to the linkage formed between two monosaccharide units through an oxygen atom by the loss of a water molecule.
- For example, in a sucrose molecule, two monosaccharide units, α- glucose and β- fructose, are joined together by a glycosidic linkage.
Question 14. What is glycogen? How is it different from starch?
Answer:
Glycogen is a carbohydrate (polysaccharide). In animals, carbohydrates are stored as glycogen. Starch is a carbohydrate consisting of two components – amylose (15-20%) and amylopectin (80-85%). However, glycogen consists of only one component whose structure is similar to amylopectin. Also, glycogen is more branched than amylopectin.
Question 15. What are the hydrolysis products of (1) sucrose and (2) lactose?
Answer: On hydrolysis, sucrose gives one molecule of α-D glucose and one molecule of β-D-fructose.
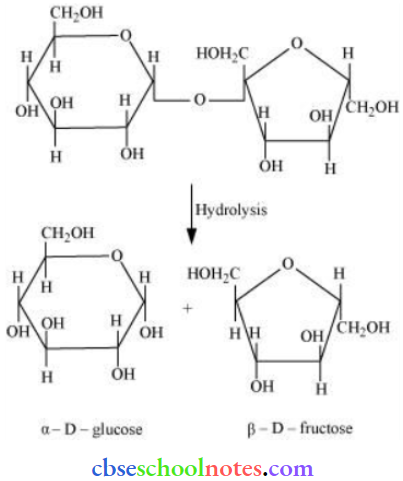
The hydrolysis of lactose gives β-D-galactose and β-D-glucose.
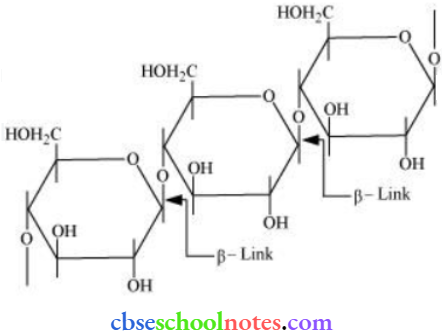
Question 16. What is the basic structural difference between starch and cellulose?
Answer:
Starch consists of two components – amylose and atm lopectin. Amylose is a long linear chain of α-D-(+)-glucose units joined by C1– C4 glycosidic linkage (α-link).
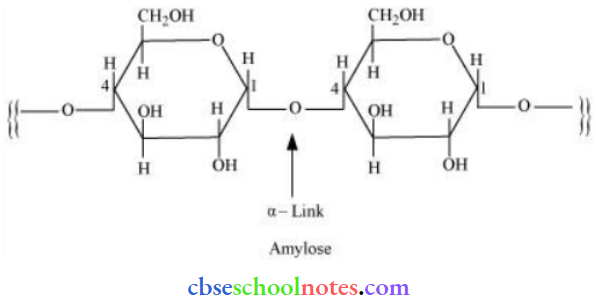
Amylopectin is a branched-chain polymer of α—D—glucose units, in which the chain is formed by C1 – C4 glycosidic linkage and the branching occurs by C1 – C6, glycosidic linkage.

On the other hand, cellulose is a straight-chain polysaccharide of β-D-glucose units joined by C1 – C4 glycosidic linkage (β-link).
Question 17. What happens when D-glucose is treated with the following reagents?
- HI
- Bromine water
- HNO3
Answer:
When D-glucose is heated with III for a long time, n-hexane is formed.
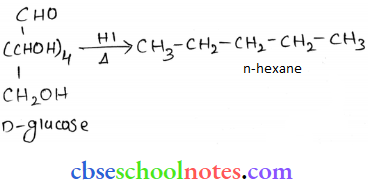
When D-glucose is treated with Br2 water, D-gluonic acid is produced.
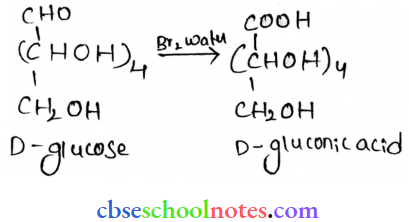
On being treated with HNO3, D-glucose gets oxidised to give saeeliaric acid.
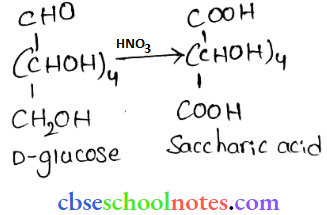
Question 18. Enumerate the reactions of D-glucose which cannot be explained by its open chain structure.
Answer:
- Aldehydes give 2, 4-DNP test. Schiff’s test, and react with NaHSO4 to form the hydrogen sulphite addition product. However, glucose does not undergo these reactions.
- The pentaacetate of glucose does not react with hydroxylamine. This indicates that a free-CHO group is absent from glucose.
- Glucose exists in two crystalline forms -α and β. The α-form (m.p. 419 K) crystallises from a concentrated solution of glucose at 303 K and the β-form (m.p.=423 K) crystallises from a hot and saturated aqueous solution at 371 K. This behaviour cannot be explained by the open chain structure of glucose.
Question 19. What are essential and non-essential amino acids? Give two examples of each type.
Answer:
Essential amino acids are required by the human body, but they cannot be synthesised in the body. They must be taken through food. For example: valine and leucine Non-essential amino acids are also required by the human body, but they can be synthesised in the body.
For example: Glycine, and Alanine
- Peptide linkage
- Primary structure
- Denaturation
Question 20. Define the following as related to proteins
- Peptide linkage
- Primary structure
- Denaturation
Answer:
Peptide linkage: The amide for pied between the -COOH group of one molecule of an amino acid and the -NH2, group of another molecule of the amino acid by the elimination of a water molecule is called a peptide linkage.
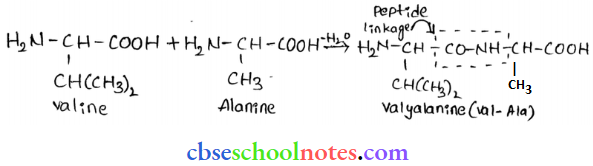
Primary structure: The primary structure of a protein refers to the specific sequence in which various amino acids are present in it. i.e. the sequence of linkage between amino acids in a polypeptide chain. The sequence in which amino acids are arranged is different in each protein. A change in the sequence creates a different protein.
Denaturation: In a biological system, a protein is found to have a unique 3- dimensional structure and a unique biological activity. In such a situation, the protein is called native protein.
- However, when the native protein is subjected to physical changes such as changes in temperature or chemical changes such as changes in pH. its H-bonds are disturbed.
- This disturbance unfolds the globules and uncoils the helix. As a result, the protein loses its biological activity. This loss of biological activity by the protein is called denaturation.
- During denaturation, the secondary and the tertiary structures of the protein get destroyed, but the primary structure remains unaltered. One of the examples of denaturation of proteins is the coagulation of egg white when an egg is boiled.
Question 21. What are the common types of secondary structures of proteins?
Answer:
There are two common types of secondary .structure of proteins:
- α- helix structure
- β-pleated sheet structure
α- Helix Structure: In this structure, the Nil group of an amino acid residue forms H-bonds with the group of the adjacent turn of the right-handed screw (α-helix).
β-pleated Sheet Structure: This structure is called so because it looks like the pleated folds of draper). In this structure, all the peptide chains are stretched out to nearly the maximum extension and then laid side by side. These peptide chains are held together by intermolecular hydrogen bonds.
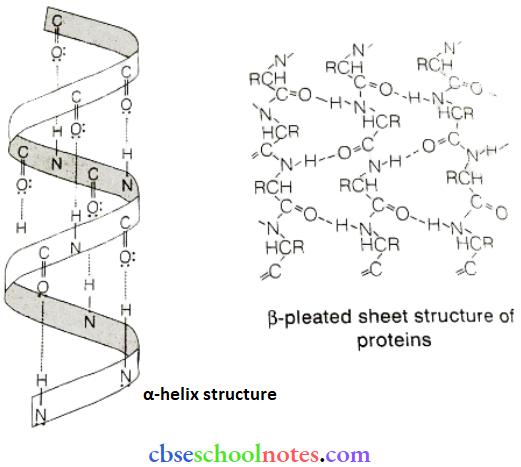
Question 22. What type of bonding helps in stabilising the a -helix structure of proteins?
Answer: The H-bonds formed between the -Ml group of each amino acid residue and the ![]() group of
group of
the adjacent turns of the α-helix help in stabilising the helix.
Question 23. Differentiate between globular and fibrous proteins.
Answer:

Question 24. How do you explain the amphoteric behaviour of amino acids?
Answer: In an aqueous solution, the carboxyl group of an amino acid can lose a proton and the amino group can accept a proton to give a dipolar ion known as a zwitter ion.
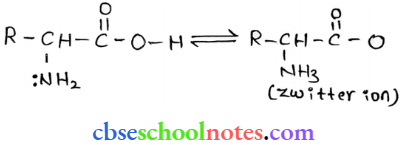
Therefore, in Zwitter ionic form, the amino acid can act both as an acid and as a base.
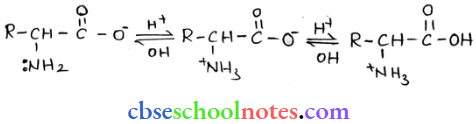
Thus, amino acids show amphoteric behaviour.
Question 25. What are enzymes?
Answer:
- Enzymes are proteins that catalyse biological reactions. They are very specific and catalyse only a particular reaction for a particular substrate. Enzymes are usually named after a particular substrate or class of substrate and sometimes after a particular reaction.
- For example, enzymes used to catalyse the hydrolysis of maltose into glucose are named as maltase.
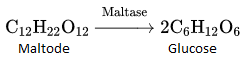
Again, the enzymes used to catalyse the oxidation of one substrate with the simultaneous reduction of another substrate are named oxidoreductase enzymes, the name of an enzyme ends with ’-ase’.
Question 26. What is the effect of denaturation on the structure of proteins?
Answer:
- As a result of denaturation. globules get unfolded and helixes get uncoiled. Secondary and tertiary structures of protein are destroyed, but the primary structures remain unaltered. It can be said that during denaturation.
- secondary and tertiary-structured proteins get converted into primary-structured proteins. Also, as the secondary and tertiary structures of a protein are destroyed, the enzyme loses its activity.
Question 27. How are vitamins classified? Name the vitamin responsible for the coagulation of blood.
Answer:
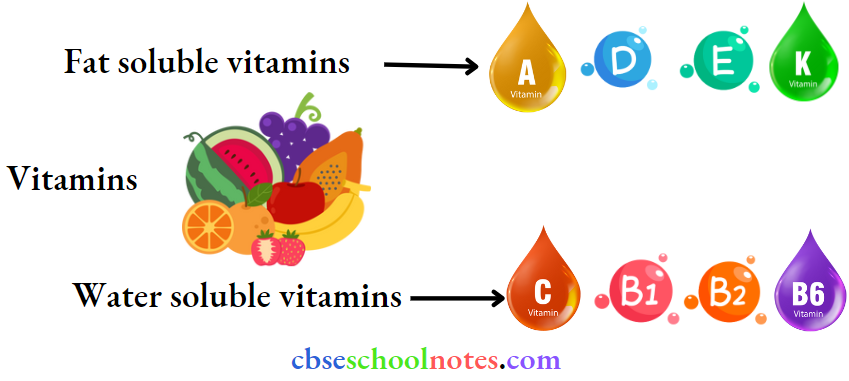
Based on their solubility in water or fat. vitamins are classified into two groups.
- Fat-soluble Vitamins: Vitamins that are soluble in fat and oils, but not in ‘water, belong to this group.
- For example: Vitamins A, D, E, and K
- Water-soluble Vitamins: Vitamins that are soluble in water belong to this group,
- For example: B group vitamins (B1, B2, B6, B12, etc.) and vitamin C
However, biotin or vitamin H is neither soluble in water nor fat. Vitamin K is responsible for the coagulation of blood.
Question 28. Why are vitamin A and vitamin C essential to us? Give their important sources,
Answer:
- The deficiency of vitamin A leads to xerophthalmia (hardening of the cornea of the eye) and night blindness. The deficiency of vitamin C leads to scurvy (bleeding gums).
- The source of vitamin A is fish liver oil, carrots, butter, and milk. The sources of vitamin C are citrus fruits. amla, and green leafy vegetables.
Question 29. What are nucleic acids? Mention their two important functions.
Answer:
Nucleic acids are biomolecules found in the nuclei of all living cells, as one of the constituents of chromosomes. There are mainly two types of nucleic acids – deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). Nucleic acids are also known as polynucleotides as they are long-chain polymers of nucleotides.
Two main functions of nucleic acids are:
- DNA is responsible for the transmission of inherent characteristics from one generation to the next. This process of transmission is called heredity.
- Nucleic acids (both DNA and RNA) are responsible for protein synthesis in a cell, even though the proteins are synthesised by the various RNA molecules in a cell, the message for the synthesis of a particular protein is present in DNA.
Question 30. What is the difference between a nucleoside and a nucleotide?
Answer:
A nucleoside is formed by the attachment of a base to the 1′ position of the sugar.
Nucleoside = Sugar + Base
On the other hand, all three basic components of nucleic acids (i.e. pentose sugar, phosphoric acid, and base) are present in a nucleotide.
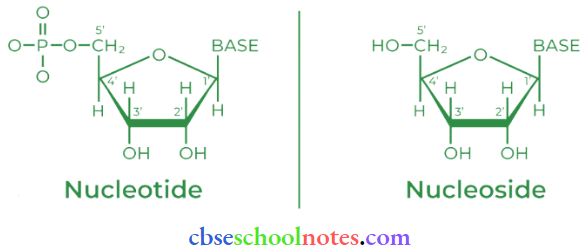
Question 31. The two strands in DNA are not identical bill are complementary. Explain.
Answer:
In the helical structure of DNA. the two strands are held together by hydrogen bonds between specific pairs of bases. Cytosin forms hydrogen bonds with guanine, while adenine forms hydrogen bonds with thymine. As a result, the two strands are complementary to each other.
Question 32. Write the important structure and functional difference between DNA and RNA.
Answer: The structural differences between DNA and RNA are as follows:
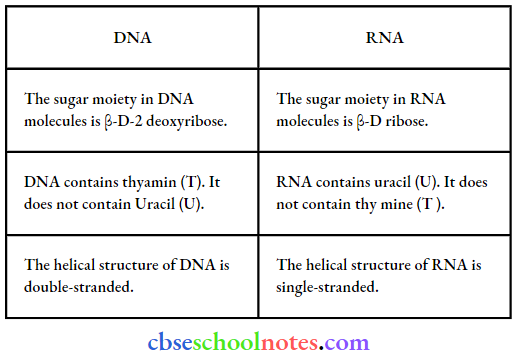
The functional differences between DNA and RNA are as follows:

Question 33. What are the different types of RNA found in the cell?
Answer:
- Messenger RNA (m-RNA)
- Ribosomal RNA (rRNA)
- Transfer RNA (t-RNA)
Question 34. What are hormones and write to its functions?
Answer:
Hormones are molecules that act as intercellular messengers. These are produced by endocrine glands in the body and are poured directly into the bloodstream which transports them to the site of action.
In terms of chemical nature, some of these are steroids, for example., estrogens and androgens: some are poly peptides for example insulin and endorphins and some others are amino acid derivatives such as epinephrine and norepinephrine.
- Insulin keeps the blood glucose level within a narrow limit. Hormone glucagon tends to increase the glucose level in the blood. The two hormones together regulate the glucose level in the blood.
- Epinephrine and norepinephrine mediate responses to external stimuli. Growth hormones and sex hormones play a role in growth and development. Thyroxine produced in the thyroid gland is an iodinated derivative of the amino acid tyrosine.
- Steroid hormones are produced by the adrenal cortex and gonads (testes in males and ovaries in females).
- Glucocorticoids control carbohydrate metabolism, modulate inflammatory reactions and are involved in reactions to stress.
- Testosterone is the major sex hormone produced in males. It is responsible for the development of secondary male characteristics (deep voice, facial hair, general physical constitution) and estradiol is the main female sex hormone. It’s responsible for the development of secondary female characteristics and participates in the control of the menstrual cycle.
- Progesterone is responsible for preparing the uterus for the implantation of a fertilised egg.
