CBSE Class 12 Physics Chapter 12 Atom Multiple Choice Questions And Answers

Question 1. The radius of the second orbit in a hydrogen atom is R. What is the radius in the third orbit?
- 3R
- 2.25 R
- 9R
- R/3
Answer: 2. 2.25 R
Read and Learn More Important Questions for Class 12 Physics with Answers
W.k.t Radius is nth orbit
rn = n2R’ → (1)
R = 4R’ → (2) [For 2 Orbit, n= 2]
r3 = 9R’ → (3) [For 2 Orbit, n= 3]
= \(9 \times \frac{R}{4}\)
∴ r3 = 2.25 R
Question 2. The wavelength of the first line of the Lyman series is X. The wavelength of the first line in the Balmer series is ______.
- \(\frac{27}{5} \lambda\)
- \(\frac{5}{27} \lambda\)
- \(\frac{9}{2} \lambda\)
- \(\frac{2}{5} \lambda\)
Answer: 1. \(\frac{27}{5} \lambda\)
⇒ \(\frac{1}{\lambda}=\mathrm{R}\left(\frac{1}{n_1^2}-\frac{1}{n_2^2}\right)\)
⇒ \(\frac{1}{\lambda}=\mathrm{R}\left(\frac{1}{1}-\frac{1}{2^2}\right)=\frac{3}{4} \mathrm{R} \Rightarrow \mathrm{R}=\frac{4}{3} \lambda\)
So, for Balmer series (n1 = 2, n2 = 3)
⇒ \(\frac{1}{\lambda^{\prime}}=\mathrm{R}\left(\frac{1}{4}-\frac{1}{9}\right)\)
⇒ \(\frac{1}{\lambda^{\prime}}=\frac{4}{3 \lambda}\left(\frac{5}{36}\right)\)
∴ \(\lambda^{\prime}=\frac{27 \lambda}{5}\)
Question 3. For the first orbit of hydrogen atom the minimum excitation potential is ______ V.
- 13.6
- 3.4
- 10.2
- 3.6
Answer: 3. 10.2
Question 4. An electron with an energy of 12.09 eV strikes a hydrogen atom In the ground state and gives its all energy to the hydrogen atom. Therefore hydrogen atoms are excited to _______ stale.
- Fourth
- third
- Second
- First
Answer: 3. Second
⇒ \(E_n=-\frac{13.6}{n^2}\)
Toral energy gained by hydrogen atoms is
-13.6+12.09 =-1.51 eV
So, \(n^2=\frac{-13.6}{-1.51}=9 \Rightarrow n=3\)
So, second excited state.
Question 5. The ratio of energies of electrons in a second excited state to the first excited slate in H-atom:
- 1:4
- 9:4
- 4:9
- 4:4
Answer: 3. 4:9
⇒ \(E_n=-13.6 \frac{z^2}{n^2}\) (used it)
For the second excited state n = 3 and the first excited state n = 2
∴ \(\frac{E_2}{E_1}=\frac{4}{9}\)
Question 6. For the first orbit of the hydrogen atom, the minimum excitation potential is ______V.
- 13.6
- 3.4
- 10.2
- 3.6
Answer: 3. 10.2
Question 7. If the potential energy of the electron in the hydrogen atom is \(\frac{-e^2}{4 \pi \varepsilon_0 r}\), then what is its kinetic energy?
- \(\frac{\mathrm{e}^2}{8 \pi \varepsilon_0 \mathrm{r}}\)
- \(\frac{-e^2}{4 \pi \varepsilon_0 r}\)
- \(\frac{-e^2}{8 \pi \varepsilon_0 r}\)
- \(\frac{e^2}{4 \pi \varepsilon_0 r}\)
Answer: 1. \(\frac{\mathrm{e}^2}{8 \pi \varepsilon_0 \mathrm{r}}\)
⇒ \(k_n=-\frac{U n}{2}\)
∴ \(k_n=+\frac{e^2}{8 \pi \varepsilon_0 r}\)
Question 8. What is the angular momentum of an electron of Li-alom in n = 5 orbit?
- 6.625 x 10-34 Js
- 5.27 x 10-34 Js
- 1.325 x 10-34 Js
- 16.56 x 10-34 Js
Answer: 2. 5.27 x 10-34 Js
⇒ \(\mathrm{mvr}=\frac{\mathrm{nh}}{2 \pi}\)
⇒ \(\frac{5 \times 6.62 \times 10^{34}}{2 \times 3.14}\)
∴ 5.27 x 10-34 Js
Question 9. A hydrogen atom absorbs 12.1 eV of energy and gets excited to a higher energy level. How many photons are emitted during the downward transition? Assume during each downward transition, one photon is emitted.
- 2 or 3
- 1 or 3
- 1 or 2
- 5 or more
Answer: 3. 1 or 2
Question 10. 13.6 eV energy is required to separate a hydrogen atom into a proton and an electron. Compute the orbital radius of the corresponding electron.
- 5.3 x 10-11 m
- 10.6 x 10-11 m
- 2.65 x 10-11 m
- 1.33 x 10-11 m
Answer: 1. 5.3 x 10-11 m
⇒ \(\mathrm{E}=-\frac{\mathrm{e}^2}{8 \pi \varepsilon_0 \mathrm{r}}\)
⇒ \(r=-\frac{e^2}{8 \pi \varepsilon_0 E}\)
By solving r = 5.29 x 10-11 m
or
⇒ \(r_1=0.529 \frac{n^2}{Z} Å\)
∴ 0.529 A
Question 11. What is the shortest wavelength present in the Paschcn scries of spectral lines?
- 821 nm
- 6563 A
- 911 nm
- 656 mm
Answer: 1. 821 nm
⇒ \(\frac{1}{\lambda}=R\left(\frac{1}{n_1^2}-\frac{1}{n_2^2}\right)\)
⇒ \(\frac{1}{\lambda}=1.097 \times 10^7\left(\frac{1}{9}-\frac{1}{\infty}\right)\)
λ = 8.21 x 10-7 m
λ = 821 x nm
Question 12. The total energy and kinetic energy of an electron in hydrogen atom arc E and K respectively.
- K = 2 E
- K = -E
- \(K=\frac{E}{2}\)
- K = E
Answer: 2. K = -E
Question 13. The ionization energy of an electron in the third excited state for a hydrogen atom is _______ eV.
- 13.6
- 1.51
- 0.85
- 3.4
Answer: 3. 0.85
⇒ \(E_n=-\frac{13.6}{n^2}\)
energy of the third excited state
⇒ \(-\frac{13.6}{16}=-0.8 .5 \mathrm{eV}\)
So Ionisation energy
= + 0.85 eV
Question 14. In the Geiger-Marsden scattering experiment, thin gold foil is used to scatter alpha particles because alpha particles will
- Not suffer more than one scattering and the gold nucleus is 50 times heavier than the alpha particle.
- Not suffer more than one scattering and the gold nucleus is lighter than the alpha particle.
- Not suffer more than a few scattering and the gold nucleus is 25 times heavier than the alpha particle.
- Suffer more than one scattering and the gold nucleus is 25 times heavier than an alpha particle.
Answer: 1. Not suffer more than one scattering; the gold nucleus is 50 times heavier than the alpha particle.
Atoms Assertion And Reason
For question numbers 1 to 5 two statements are given-one labelled Assertion (A) and the other labelled Reason (R). Select the correct answer to these questions from the codes (1), (2), (3), and (4) as given below.
- Both A and R are true and R is the correct explanation of A
- Both A and R are true but R is NOT the correct explanation of A
- A is true but R is false
- A is false and R is also false
Question 1. Assertion: The mass of the atom is concentrated in the nucleus.
Reason: The mass of a nucleus can be either less than or more than the sum of the masses of nucleons present in it.
Answer: 3. A is true but R is false
Question 2. Assertion: Bohr’s orbits are also called stationary stales.
Reason: In Bohr’s orbits electron revolves in a fixed path.
Answer: 1. Both A and R are true and R is the correct explanation of A
Question 3. Assertion: A hydrogen atom consists of only one electron but its emission spectrum has many lines.
Reason: The Lyman series is found in the emission spectrum.
Answer: 2. Both A and R are true but R is NOT the correct explanation of A
Question 4. Assertion: For the scattering of a-particles at a large angle only the nucleus of the atom is responsible.
Reason: Nucleus is very heavy in comparison to electrons.
Answer: 1. Both A and R are true and R is the correct explanation of A
Question 5. Assertion: Atom arc is not electrically neutral.
Reason: The number of protons and electrons is different
Answer: 4. A is false and R is also false
CBSE Class 12 Physics Chapter 12 Atoms Short Questions And Answers
Question 1. Show that the radius of the orbit in a hydrogen atom varies as n2 where n is the principal quantum number of the atom.
Answer:
Coulomb force: The electrostatic attraction force between the electron and nucleus is _______
⇒ \(\mathrm{F}_{\mathrm{e}}=\frac{\mathrm{kZe}^2}{\mathrm{r}_{\mathrm{n}}^2}\) → (1) \(\left\{\mathrm{F}_{\mathrm{e}}=\frac{\mathrm{k} \mathrm{q}_1 \mathrm{q}_2}{\mathrm{r}^2}\right.\)
Centripetal Force = \(\frac{m v_n^2}{r_n}\)
In equilibrium \(\frac{m v_n^2}{r_n}=\frac{k Z e^2}{r_n^2}\)
⇒ \(\mathrm{v}_{\mathrm{n}}^2=\frac{\mathrm{kZe^{2 }}}{\mathrm{mr}_{\mathrm{n}}}\) → (2)
Then form Bohr’s second postulate \(m v_n r_n=\frac{n h}{2 \pi}\)
∴ \(\mathrm{v}_{\mathrm{n}}=\frac{\mathrm{nh}}{2 \pi \mathrm{mr}_{\mathrm{n}}}\) → (3)
From eq (2) and (3)
⇒ \(\frac{n^2 h^2}{4 \pi^2 m^2 r_n^2}=\frac{k Z e^2}{m r_n}\)
⇒ \(r_n=\left(\frac{h^2}{4 \pi^2 e^2 k m}\right) \frac{n^2}{Z}\)
Put the value, π, k, h, m, e
∴ \(r_n=0.529 \times 10^{-8} \times \frac{n^2}{Z} \mathrm{~cm}=0.529 \times \frac{n^2}{Z}Å\)
Question 2. How does one explain, Bohr’s second postulate of quantization of orbital angular momentum using the de Broglie’s hypothesis?
Answer:
The behavior of particle waves can be viewed as analogous to the waves traveling on a siring. Particle waves can lead to standing waves held under resonant conditions. When a stationary string is plucked, several wavelengths are excited but only those wavelengths survive which form a standing wave in the string.
Thus, in a string standing waves arc formed only when the total distance traveled by f a wave is an integral number of wavelengths. Hence, for any electron moving in a circular orbit of radius rn, the total distance is equal to the circumference of the orbit. 2πrn .
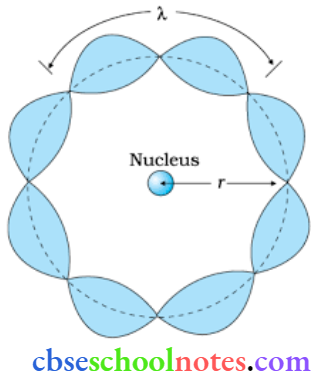
2πrn = n → (1)
Here, λ is de Broglie wavelength.
We know
λ = h/p
or λ = h/mvn → (2)
Where mvn is the momentum of an electron revolving in the nth orbit,
From equation (2) and equation (1) we get.
2πrn = nh/mvn
L = mvn rn = nh/2π
Hence, de Broglie’s hypothesis successfully proves Bohr’s second postulate.
Question 3. Given the value of the ground state energy of hydrogen atom as -13.6 eV, find out its kinetic and potential energy in the ground and second excited states.
Answer:
Ground stale energy of hydrogen atom as -13.6 eV,
En = Total energy = \(-\frac{13.6 \mathrm{eV}}{n^2}\)
K.e = -T.E and P.e = 2 T.E
For ground stale n = 1, then
⇒ \(\mathrm{T} . \mathrm{E} .=\frac{-13.6 \mathrm{eV}}{(1)^2}=-13.6 \mathrm{eV}\)
K.E. = +13.6 eV
P.E. = -27.2 eV
For second excited state n = 3, then
∴ \(\text { T.E. }=\frac{-13.6}{9}=-1.51 \mathrm{eV}\)
K.E. = +1.51 eV
P.E. = -3.02 eV
Question 4. Given the ground stale energy E0 = -13.6 cV and Bohr radius r0 = 0.53 Å. Find out how the dc Broglie wavelength associated with the electron orbiting in the ground stale would change when it jumps into the first excited state.
Answer:
Given ground slate energy E0 = – 13.6 eV
Energy in the first excited state = \(E_1=\frac{-13.6 \mathrm{eV}}{(2)^2}=-3.4 \mathrm{eV}\)
∴ \(\lambda=\frac{h}{\sqrt{2 \mathrm{mE}_1}}=\frac{6.63 \times 10^{-34}}{\sqrt{2 \times\left(9.1 \times 10^{-31}\right)\left(3.4 \times 1.6 \times 10^{-19}\right)}}\)
∴ \(\lambda=\frac{6.63 \times 10^{-34}}{9.95 \times 10^{25}}=6.66 \times 10^{-10} \mathrm{~m} \simeq 6.7 \times 10^{-10} \mathrm{~m}\)
Question 5. A hydrogen atom initially in the ground slate absorbs a photon with energy 12.5 eV. Calculate the longest wavelength of the radiation emitted and identify the series to which it belongs.
Answer:
We Know, \(\frac{1}{\lambda_{\max }}=R\left(\frac{1}{n_f^2}-\frac{1}{n_i^2}\right)\)
The energy of the incident photon = 12.5 eV
Energy of ground stale = -13.6 eV
∴ The energy of the hydrogen atom after absorption of a photon can be -1.1 eV
This means that electrons can go to the excited state ni = 3. Now it emits photons of maximum wavelength on going to nf = 2
⇒ \(\frac{1}{\lambda_{\max }}=\mathrm{R}\left\{\frac{1}{2^2}-\frac{1}{3^2}\right\}\)
⇒ \(\lambda_{\text {mat }}=\frac{36}{5 \mathrm{R}}=\frac{36}{5 \times 1.1 \times 10^7}=6.545 \times 10^{-7} \mathrm{~m}=6.545 \mathrm{~A}^{\circ}\)
It belongs to the Balmer Series.
Question 6. When is the Hα line in the emission spectrum of the hydrogen atom for the Balmer series obtained? Calculate the frequency of the photon emitted during this transition.
Answer:
Hα is a specific deep-red visible spectral line in the Balmer series with a wavelength of 656.28 nm, it occurs when a hydrogen electron transits from its 3rd to 2nd lowest energy level.
This transition produces an H-alpha photon and the 1st line of the Balmer series.
⇒ \(\frac{1}{\lambda}=R\left[\frac{1}{n_f^2}-\frac{1}{n_i^2}\right]\)
⇒ \(\mathrm{R}\left[\frac{1}{2^2}-\frac{1}{3^2}\right] \Rightarrow \frac{1}{\lambda}=1.097 \times 10^7\left[\frac{5}{36}\right]\)
∴ \(v=\frac{c}{\lambda}=\frac{3 \times 10^8 \times 1.097 \times 10^7 \times 5}{36}\) = 4.57 x 1014 Hz
Question 7. The energy levels of a hypothetical atom are shown below. Which of the shown transitions will result in the emission of a photon of wavelength 275 nm?
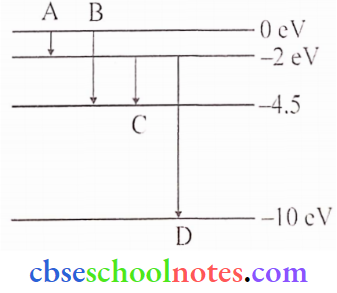
Answer:
Using relation, \(\mathrm{E}=\frac{1242 \mathrm{eV}}{\lambda(\text { in } \mathrm{nm})}\)
Here, = 275nm, \(\mathrm{E}=\frac{1242}{275}=4.5 \mathrm{eV}\)
This energy of photon exists corresponding to ‘B’.
Question 8. State Bohr’s postulate of hydrogen atom which successfully explains the emission lines in the spectrum of hydrogen atom.
Use the Rydberg formula to determine the wavelength of the line.
[Given : Rydberg constant R = 1.03 x 107 m-1]
Answer:
According to Bohr’s postulate when an e– jumps from one orbit to another, the energy difference between them is emitted in the form of energy i.e. as a photon which shows the emission lines in the spectrum of hydrogen atoms.
⇒ \(E_i-E_f=h \nu=\frac{h c}{\lambda}\)
Now, the Rydberg formula for the Balmer series is
⇒ \(\frac{1}{\lambda}=R\left(\frac{1}{n_f^2}-\frac{1}{n_i^2}\right)\) where R = Rydberg constant = 1 .09 x 107 m-1
The Hα– line of the Balmer series is obtained when an e jumps to the second orbit (nf = 2) from
the third orbit (n. = 3).
Further, \(\frac{1}{\lambda}=1.03 \times 10^7\left(\frac{1}{2^2}-\frac{1}{3^2}\right)=6.99 \times 10^{-7}=699 \mathrm{~nm}\)
‘λ’ lies in the visible region.
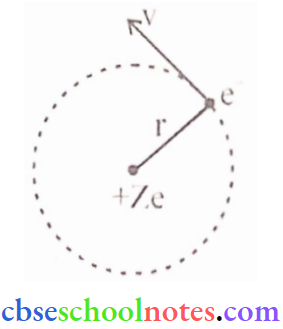
Question 9. Using Rutherford’s model of the atom, derive the expression for the total energy of a tin electron in the hydrogen atom. What is the significance of total negative energy possessed by the electron?
Answer:
According to Rutherford’s nuclear model of the atom, the electrostatic force of attraction Fe between the revolving electron and the nucleus provides the centripetal force (Fc).
Thus, Fc = Fe
∴ \(\frac{m v^2}{r}=\frac{1}{4 \pi \varepsilon_0} \cdot \frac{e^2}{r^2}\) (∵ z = 1)
Thus the relation between the orbit radius and the e- velocity is, \(\mathrm{r}=\frac{\mathrm{e}^2}{4 \pi \varepsilon_0 \mathrm{mv}^2}\)
The K.E. (K) and electrostatic potential energy (U) of electrons in hydrogen atoms are
⇒ \(K=\frac{1}{2} m v^2=\frac{e^2}{8 \pi \varepsilon_0 r}\)
and \(\mathrm{U}=\frac{-\mathrm{e}^2}{4 \pi \varepsilon_0 \mathrm{r}}\) (-ve sign shows, that the nature of force is attractive)
Thus, the total rnech. energy E of an e– is,
∴ \(\mathrm{E}=\mathrm{K}+\mathrm{U}=\frac{\mathrm{e}^2}{8 \pi \varepsilon_0 \mathrm{r}}-\frac{\mathrm{e}^2}{4 \pi \varepsilon_0 \mathrm{r}}=\frac{-\mathrm{e}^2}{8 \pi \varepsilon_0 \mathrm{r}}\)
Total energy (E) is -ve which shows that e– is bound to the nucleus, if (E) were +ve then e- would leave the the atom.
Question 10.
- Stale Bohr’s postulate of the hydrogen atom gives the relationship to the frequency of emitted photons in a transition.
- An electron jumps from the fourth to the first orbit in an atom. How many maximum number of spectral lines can be emitted by the atom? To which series do these lines correspond?
Answer:
1. According to Bohr’s theory, energy is quantized i.c. for each orbital the corresponding energy is
given as
ΔE = hv
2. There will be 6 spectral lines and they correspond to.
Max. number of spectral lines = \(\frac{n(n-1)}{2}\)
For the fourth orbit n = 4
Then, Max. possible lines = \(\frac{4(4-1)}{2}\)
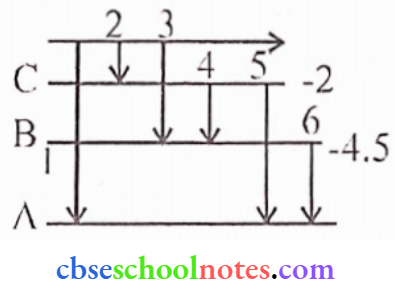
A-Lyman Series, B-Balmcr series, C-Paschcn series
Question 11. A 12.5 eV electron beam is used to excite a gaseous hydrogen atom at room temperature. Determine the wavelengths and the corresponding series of the lines emitted.
Answer:
When an electron beam of 12.5 eV is used to excite hydrogen, then the probable transition is from n=1 to n=3 (i.e. E3-E1 = -1.5l-(-l 3.6) = 12.09e V)
On de-excitation, the electron may jump by following the ways
3 → I, 3 → 2, 2 → 1
Possible wavelength and their corresponding series of lines emitted are
1. From n=3 to n = 1, \(\frac{1}{\lambda}=R\left[\frac{1}{1^2}-\frac{1}{3^2}\right] \Rightarrow \frac{1}{\lambda}=R\left[1-\frac{1}{9}\right]=R\left(\frac{8}{9}\right)\)
or \(\lambda=\frac{9}{8 \mathrm{R}}=1.026 \times 10^{-7} \mathrm{~m}=102.6 \mathrm{~nm}\)
It belongs to the Lyman series.
2. From n = 3 to n = 2,
⇒ \(\frac{1}{\lambda}=R\left[\frac{1}{2^2}-\frac{1}{3^2}\right]=R\left[\frac{1}{4}-\frac{1}{9}\right] \Rightarrow \frac{1}{\lambda}=R\left[\frac{5}{36}\right]\)
∴ \(\lambda=\frac{36}{5 \mathrm{R}}=6.563 \times 10^{-7} \mathrm{~m}=656.3 \mathrm{~nm}\)
It belongs to the Balmer series.
3. From n = 2 to n = 1,
⇒ \(\frac{1}{\lambda}=R\left[\frac{1}{1^2}-\frac{1}{2^2}\right]=R\left[1-\frac{1}{4}\right] \Rightarrow \frac{1}{\lambda}=\frac{3 R}{4}\)
A = 1.215 x 1 0-7m = 121.5 nm
It belongs to the Lyman series.
Question 12.
- In A Geiger-Marsden Experiment. Find the distance of the closest approach to the gold nucleus (mass no. = 79)m of a 7.7 Me V α-particle before it comes momentarily to rest and reverses its direction.
- Plot a graph between several scattered α-particles detected in the gold foil experiment and the angle of scattering. What is the main assumption in plotting this graph?
Answer:
K.E. = P.E
⇒ \(K E_{\alpha}=\frac{k q_1 q_2}{r}\) where q1 = 79e, q2 = 2e
So r = \(\mathrm{r}=\frac{\mathrm{kq}_1 \mathrm{q}_2}{\mathrm{KE}_\alpha}=\frac{9 \times 10^9 \times\left(79 \times 1.6 \times 10^{-19}\right)\left(2 \times 1.6 \times 10^{-19}\right)}{7.7 \times 10^6 \times 1.6 \times 10^{-19}}=\frac{18 \times 79 \times 1.6}{7.7} \times 10^{-16} \mathrm{~m}\)
r = 295.4 x 10-16 = 29.54 m = 30 fm
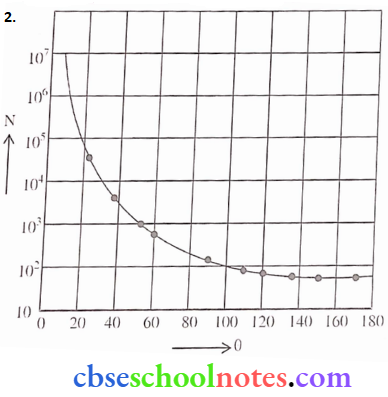
0 = Scattering angle
N= No. of α-Particles
The plot is Schematic and not according to scale.
Question 13.
- state Bohr’s quantization condition for defining stationary orbits. How does de Broglie’s hypothesis explain the stationary orbits?
- Find the relation between the three wavelengths λ1, λ2, λ3 and from the energy level diagram shown below.
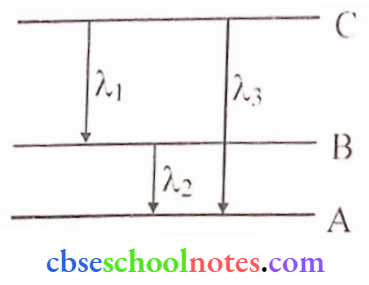
Answer:
1. Bohr’s quantization condition: The electron can revolve around the nucleus only in those circular orbits in which the angular momentum of an electron is an integral multiple of \(\frac{h}{2 \pi}\), where h is Planck’s constant. \(\mathrm{mvr}=n \frac{\mathrm{h}}{2 \pi}\) [m = mass, v = velocity of electron]
Their circular orbits are stationary, de Broglie hypothesis: de-Broglie interprets Bohr’s 2nd postulate in terms of the wave nature of the electron. According to him- The electron can revolve in certain stable orbits for which the angular momentum is some integral multiple of h/2π.
Mathematically, \(2 \pi r_n=\frac{n h}{m v}\)
Therefore, \(\mathrm{mvr}_{\mathrm{n}}=\frac{\mathrm{nh}}{2 \pi}\)
2. \(E_{C B}=\frac{h c}{\lambda_1} \Rightarrow E_{B A}=\frac{h c}{\lambda_2} \Rightarrow E_{C A}=\frac{h c}{\lambda_3}\)
here ECB = Energy gap between levels B and C
ECA = Energy gap between levels A and C
EBA= Energy gap between levels A and B
ECA = ECB + EBA
∴ \(\frac{h c}{\lambda_3}=\frac{h c}{\lambda_1}+\frac{h c}{\lambda_2} \Rightarrow \frac{1}{\lambda_3}=\frac{1}{\lambda_1}+\frac{1}{\lambda_2} \Rightarrow \lambda_3=\frac{\lambda_1 \lambda_2}{\lambda_1+\lambda_2}\)
Question 14. State Bohr’s postulate to explain stable orbits in a hydrogen atom. Prove that the speed with which the electron revolves in nlh orbit is proportional to (1/n).
Answer:
Bohr’s postulate:
Electron revolves around the nucleus in those orbits for which the angular momentum is an
integral multiple of h/2π.
∴ \(\mathrm{mvr}=\frac{\mathrm{nh}}{2 \pi}\)
Speed of e– in nth orbit
For hydrogen radius of nth orbit is given by = \(r_n=\frac{\varepsilon_0 n^2 h^2}{\pi m c^2}\)
Form Bohr’s postulate
⇒ \(m v_n r_n=\frac{n h}{2 \pi}\)
⇒ \(m v_n\left(\frac{\varepsilon_0 n^2 h^2}{\pi m e^2}\right)=\frac{n h}{2 \pi}\)
∴ \(\mathrm{V}_{\mathrm{n}}=\frac{\mathrm{c}^2}{2 \varepsilon_0 h n} \Rightarrow \mathrm{V}_{\mathrm{n}} \propto \frac{1}{\mathrm{n}}\)
Question 15. A narrow beam of protons, each having 4.1 MeV energy is approaching a sheet of lead (Z = 82). Calculate :
- The speed of a proton in the beam, and
- The distance of its closest approach
Answer:
Energy of proton = 4. 1 MeV, Z = 82
1. Speed of a proton
⇒ \(\frac{1}{2} m_p v_p^2=4.1 \times 10^6 \times 1.6 \times 10^{-19}\)
∴ \(v_p^2=\frac{2 \times 4.1 \times 1.6 \times 10^{-13}}{1.67 \times 10^{-27}}=7.85 \times 10^{14}\)
Vp = 2.8 x 107 m/s
2. Distance of closest approach
⇒ \(K. E=\frac{k e(z e)}{r_0}\)
⇒\(r_0=\frac{K Z e^2}{r_0}=\frac{9 \times 10^{9} \times 82 \times\left(1.6 \times 10^{-19}\right)^2}{4.1 \times 10^6 \times 1.6 \times 10^{-19}}=288 \times 10^{-16}\)
∴r0 = 2.9 x 10-14 m-1
CBSE Class 12 Physics Chapter 12 Long Questions And Answers
Question 1. According to the third postulate of Bohr’s model, when an atom makes a transition from the higher energy stale with quantum number nj to the lower energy stale with quantum number nf(nf <ni), the difference of the energy is carried away by the photon of frequency such that hv = Eni-Enf.
Since both ni and nf are integers, this immediately shows that in transitions between the different atomic levels, light is radiated in various discrete frequencies. For the hydrogen atom spectrum, the Balmcr formula corresponds to nf = 2 and ni = 3, 4, 5, etc. This result of Bohr’s model suggested the presence of other scries spectra for hydrogen atoms – those corresponding to the transitions resulting from nf = 1 and ni = 2, 3, etc: and nf = 3 and ni = 4, 5, etc., and so on. Such series were identified in the course of spectroscopic investigations and are known as the Lyman, Balmer, Paschen, Brackett, and Pfund series. The electronic transitions corresponding to this series are shown
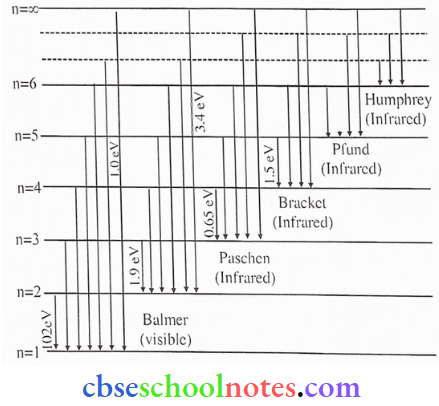
(1). The total energy of an electron in an atom in an orbit is -3.4 eV. Its kinetic and potential energies are respectively
- 3.4 eV, 3.4 eV
- -3.4 eV, -3.4 eV
- -3.4eV,-6.8 eV
- 3.4 eV, -6.8 eV
Answer: 4. 3.4 eV, -6.8 eV
(2). Given the value of the Rydberg constant is 107 m-1, the wave number of the last line of the Balmer series in the hydrogen spectrum will be
- 0.5 x 107 m-1
- 0.25 x 107 m-1
- 2.5 x 107 m-1
- 0.025 x 104 m-1
Answer: 2. 0.25 x 107 m-1
(3). The ratio of the wavelength of the last line of Balmcr scries and the last line of the Lyman series
- 0.5
- 2
- 1
- 4
Answer: 4. 1
(4). The wavelength of Balmer scries lies in
- Ultraviolet region
- Infrared region
- Far infra-red region
- Visible region
Answer: 4. Visible region
Question 2. Neutrons mid protons tire identical particles in the sense that their masses are nearly the same and the force called nuclear force, does not distinguish between them, Nuclear force is the strongest force. The stability of the nucleus is determined by the neutron Proton ratio or mass defect or packing fraction, The Shape of the nucleus is calculated by quadruple moment and the spin of the nucleus depends on an even or odd mass number. The volume of the nucleus depends on the mass number. The whole mass of the atom (nearly 99%) is centered at the nucleus.
(1). The correct statement about the nuclear force is as follows:
- Charge independent
- Short range force
- Nonconservative force
- All of these
Answer: 4. All of these
(2). The range of a nuclear force is the order of:
- 2 x 10-10 m
- 1.5 x 10-20 m
- 1.2 x 10-4m
- 1.4 x 10-15 m
Answer: 4. 1.4 x 10-15 m
(4). A force between two protons is the same as the force between a proton and a neutron. The nature of the force is:
- Electrical force
- Weak nuclear force
- Gravitational force
- Strong nuclear force
Answer: 1. Electrical force
