CBSE Class 12 Physics Chapter 13 Nuclei Multiple Choice Questions And Answers
Question 1. If the radii \({ }_{13}^{27} \mathrm{Zn}\) abd \({ }_{30}^{64} \mathrm{Zn}\) nucleus are R1 and R2 respectively, Then \(\frac{R_1}{R_2}\) = ________.
- \(\frac{27}{64}\)
- \(\frac{3}{4}\)
- \(\frac{9}{16}\)
- \(\frac{13}{30}\)
Answer: 2. \(\frac{3}{4}\)
Read and Learn More Important Questions for Class 12 Physics with Answers
⇒ \(R=R_0 A^{\frac{1}{3}}\)
∴ \(\frac{R_1}{R_2}=\left(\frac{A_1}{A_2}\right)^{1 / 3}=\left(\frac{27}{64}\right)^{1 / 3}=\frac{3}{4}\)
Question 2. In the radioactive transformation \({ }_z^A X \rightarrow{ }_{Z-1}^A X_1 \rightarrow{ }_{Z-1}^{A-4} X_2 \rightarrow_Z^{A-4} X_3\) which are the successively emitted radioactive radiations?
- β–, α, β–
- α, β–, β–
- β–, β–, α
- α, α, β–
Answer: 1. β–, α, β–
Question 3. Which are the isotone, isobar and isotope nuclei respectively of \({ }_6^{12} \mathrm{C}\) from among \({ }_6^{14} \mathrm{C},{ }_5^{12} \mathrm{~B},{ }_7^{13} \mathrm{~N}\)?
- \({ }_5^{12} \text { B, }{ }_6^{14} \mathrm{C,} { }_7^{13} \mathrm{~N}\)
- \({ }_6^{14} \mathrm{C},{ }_7^{13} \mathrm{~N},{ }_5^{12} \mathrm{~B}\)
- \({ }_7^{13} \text { N, }{ }_5^{12} \mathrm{B,} { }_6^{14} \mathrm{~C}\)
- \({ }_6^{14} \mathrm{C},{ }_5^{12} \mathrm{~B,} { }_7^{13} \mathrm{~N}\)
Answer: 3. \({ }_7^{13} \text { N, }{ }_5^{12} \mathrm{B,} { }_6^{14} \mathrm{~C}\)
Question 4. The binding energy per nucleon is almost constant for the nuclei having atomic mass number ________.
- 30 < A < 240
- 30 < A < 170
- 170 < A < 230
- 156 < A < 192
Answer: 2. 30 < A < 170
Question 5. The energy equivalent to 1 gram (g) substance is _________ J.
- 9 x 107
- 9 x 1010
- 9 x 1013
- 9 x 108
Answer: 3. 9 x 1013
∴ \(E=m c^2=\frac{1}{1000} \times 9 \times 10^{16}=9 \times 10^{13} \mathrm{~J}\)
Question 6. \({ }_{80}^{198} \mathrm{Hg} \text { and }{ }_{79}^{197} \mathrm{Au}\) are example of __________.
- Isotopes
- Isomers
- Isobars
- Isotones
Answer: 4. Isotones
Question 7. In a curve of binding energy per nucleon versus mass number (A), the maximum value of Ebn is 8.75 MeV/nucleon the value of a corresponding atomic mass number is _______.
- 56
- 235
- 238
- 171
Answer: 1. 56
Question 8. The saturation property of the nuclear forces is because they are:
- Charge independent forces
- Non-central forces
- Spin-dependent forces
- Short-range forces
Answer: 4. Short-range forces
CBSE Class 12 Physics Chapter 13 Nuclei Assertion And Reason
For question numbers 1 to 5 two statements are given-one labelled Assertion (A) and the other labelled Reason (R). Select the correct answer to these questions from the codes (1), (2), (3), and (4) as given below.
- Both A and R are true and R is the correct explanation of A
- Both A and R are true but R is NOT the collected explanation of A
- A is true but R is false
- A is false and R is also false
Question 1. Assertion: Energy is released in nuclear fission.
Reason: The total binding energy of the fission fragments is larger than the total binding energy of the parent.
Answer: 1. Both A and R are true and R is the correct explanation of A
Question 2. Assertion: The nucleus may emit negatively charged particles.
Reason: The nucleus contains a negative charge also.
Answer: 3. A is true but R is false
Question 3. Assertion: Nuclear binding energy per nucleon is in the order \({ }_4^9 \mathrm{Be}>{ }_3^7 \mathrm{Li}>{ }_2^4 \mathrm{He}\)
- Reason: Binding energy per nucleon increases linearly with differences in several neutrons and protons.
- Answer: 4. A is false and R is also false
Question 4. Assertion: Isobars arc the clement having the same mass number but a different atomic number.
Reason: Neutrons and protons are present inside the nucleus.
Answer: 2. Both A and R are true but R is NOT the collected explanation of A
Question 5. Assertion: The density of all the nuclei is the same.
Reason: The radius of the nucleus is directly proportional to the cube roots of the mass number
Answer: 1. Both A and R are true and R is the correct explanation of A
CBSE Class 12 Physics Chapter 13 Nuclei Short Questions And Answers
Question 1. Obtain approximately the ratio of the nuclear radii of the gold isotope \({ }_{79} \mathrm{Au}^{197}\) and silver isotopes \({ }_{47} \mathrm{Ag}^{107}\)
Answer:
A1 = 197 and A2 = 107 R = R0A1/3
⇒ \(\frac{R_1}{R_2}=\left[\frac{A_1}{A_2}\right]^{1 / 3}=\left[\frac{197}{107}\right]^{1 / 3}=1.225\)
Question 2. The relation R = R0 A1/3. where R0 is a constant and A is the mass number of A nucleus, showing that the nuclear matter density is nearly constant. (i.e independent of A)
Answer:
Density of nucleus matter = \(\frac{\text { Mass of nucleus }}{\text { Volume of nucleus }}\)
⇒ \(\rho=\frac{\mathrm{mA}}{\frac{4}{3} \pi \mathrm{R}^3}\) [R = R A1/3]
⇒ \(\rho=\frac{3 \mathrm{~m}}{4 \pi \mathrm{R}_0^3}\) = \(\rho=\frac{\mathrm{A} \times 1.66 \times 10^{27} \mathrm{~kg}}{\frac{4}{3} \times 3.14 \times\left(1.2 \times 10^{-15}\right)^3 \mathrm{~A}} \approx 10^{17} \mathrm{~kg} / \mathrm{m}^3\)
The density of the nucleus is constant.
Question 3. Draw a graph showing the variation of the potential energy of a pair of nucleons as a function of their separation to indicate the region in which the nuclear force is (1) Attractive, (2)Repulsive
Answer:
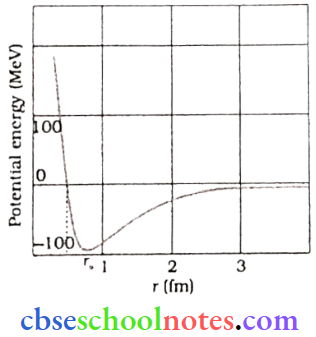
The potential energy is minimum at a distance r() of about 0.8 fm. This means that the force is attractive for distances larger than 0.8 fm and repulsive if they are separated by distances less than 0.8 fm.
Question 4. What is nuclear force? Write its specific properties.
Answer:
Nuclear Force: The nuclear force is the strongest force of nature that holds nucleons in the nucleus of an atom.
Properties: Nuclear forces act between a pair of neutrons, a pair of protons, and a neutron-proton pair with the same strength, i.e. nuclear force is charge-independent.
- The nuclear forces are very short-range forces, hence show saturation property.
- The nuclear forces are dependent on the spin angular momentum of nucleons.
- Nuclear forces arc non-central forces.
Question 5. Differentiate between nuclear fission and fusion. Give an example of each. Which of the above reactions take place in a nuclear reactor?
Answer:
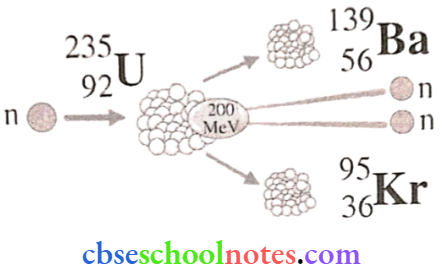
1. Nuclear Fission: Nuclear fission is the phenomenon of splitting a heavy nucleus (usually A > 230) into two or more lighter nuclei.
Example: \({ }_{92} \mathrm{U}^{235}+{ }_0 \mathrm{n}^1 \rightarrow_{92} \mathrm{U}^{236} \rightarrow_{56} \mathrm{Ba}^{141}+{ }_{36} \mathrm{Kr}^{92}+3 { }_0\mathrm{n}^1+\mathrm{Q}\)
2. Nuclear Fission: The process of combining of two lighter nuclei to form one heavy
nucleus is called nuclear fusion.
Example: \(4_1 \mathrm{H}^1 \rightarrow{ }_2 \mathrm{He}^4+2_1 \mathrm{e}^0+2 v+26.7 \mathrm{MeV}\)
A nuclear reactor is based on a controlled chain reaction.
Question 5.
- Explain the processes of nuclear fission and fusion using the plot of binding energy per nucleon (BE/A) versus the mass number A.
- Why are neutrons preferred as better projectiles in causing nuclear reactions?
Answer:
1. The value of binding energy per nucleon gives a measure of the stability of that nucleus greater the binding energy per nucleon of a nucleus, the more stable the nucleus.
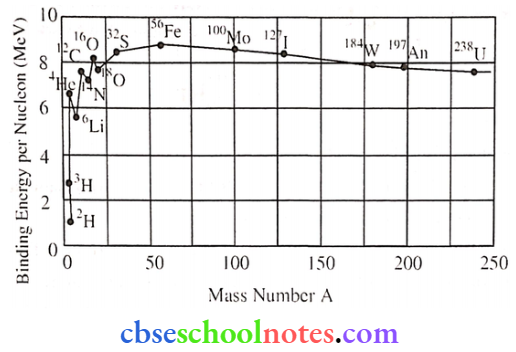
The above graph shows, the binding energy per nucleon drawn against mass number A. The saturation effect of nuclear forces is properly responsible for the approximate constancy of binding energy in the range 30 < A < 170.
It is clear from the curve that the binding energy per nucleon of the fused nuclei is more than that of the lighter nuclei taking part in nuclear fusion. Hence energy is released in this process.
In nuclear fission, the sum of the masses of the final products is less than the sum of the masses of the reactant components. Hence energy is released in this process.
2. These are uncharged particle
Question 6.
- Write the basic nuclear process involved in the emission of β+ in a symbolic form, by a radioactive nucleus.
- In the reactions given below:
- \({ }_6^{11} \mathrm{C} \rightarrow{ }_y^{z} \mathrm{B}+x+v\)
- \({ }_6^{12} \mathrm{C}+{ }_6^{12} \mathrm{C} \rightarrow{ }_{\mathrm{a}}^{20} \mathrm{Ne}+{ }_1^{\mathrm{c}} \mathrm{He}\)
Answer:
1. \({ }_z X^{A} \rightarrow{\beta^+}{ }_{z-1} Y^{A}+{ }_{+1} e^{0}+v+\text { energy }\)
In the emission of β+, mass no. (A) remains the same but atomic no. (Z) decreases by 1 unit.
2. (1). \(x \longrightarrow{ }_{+1} e^0, y \longrightarrow 5, z \longrightarrow 11\)
(2). \(\mathrm{a} \longrightarrow 10, \mathrm{~b} \longrightarrow 2, \mathrm{c} \longrightarrow 4\)
CBSE Class 12 Physics Chapter 13 Nuclei Long Questions And Answers
Question 1. When the nucleus of an atom splits into lighter nuclei through a nuclear reaction the process is termed as nuclear fission. This decay can be natural spontaneous splitting by radioactive decay or can be simulated in a lab by achieving the necessary conditions (bombarding with neutrons). The resulting fragments tend to have a combined mass that is less than the original.
(1). This splitting of a nucleus into smaller nuclei is:
- Fusion
- Fission
- Half-life
- Gamma-radiation
Answer: 2. Fission
(2). Name the moderator used in the nuclear reactor.
- Plutonium
- Thorium
- Graphite
- Berilium
Answer: 3. Graphite
(3). Which isotope of Uranium can sustain the chain reaction?
- U-230
- U-235
- U-245
- U-225
Answer: 2.U-235
(4). Which of the following atomic particles has the least mass?
- Proton
- Electron
- Deuleron
- Neutron
Answer: 2. Electron
Question 2. Uranium-235 (235U) is an isotope of uranium making up about 0.72 % of natural uranium. It is a fissile material i.c., and it can sustain a fission chain reaction. U-235 has a half-life of 703.8 million years. The main use for uranium today is for fuel in nuclear power plants. Depleted uranium is used in bullets and larger projectiles to make them hard and dense enough to reach through armored targets. Inhaling large concentrations of this radioactive element can cause lung cancer. It is also a toxic chemical, its ingestion can cause damage to kidneys, and its radioactive properties could cause cancers of bones or liver.
(1). A nuclei having the same number of neutrons but a different number of protons/atomic numbers and called:
- Isobars
- Isomers
- Isotones
- Isotopes
Answer: 3. Isotones
(2). For a nuclear fission process, suitable nuclei are:
- Any nuclei
- Heavy nuclei
- Lighter nuclei
- Nuclei lying in the middle of the periodic table
Answer: 2. Heavy nuclei
(3). Mass-energy equation was propounded by:
- Newton
- Madam Curie
- C.V. Raman
- Einstein
Answer: 4. Einstein
(4). Name the antiparticle of electron:
- Positro
- α-Particle
- proton
- β- Particle
Answer: 1. Positro
